文献精选
4.1 引言
皮肤是人体的主要器官之一。它覆盖全身,与人体其它器官密切相连,起着保护人体不受外部刺激或伤害的作用。人的皮肤从表面上来看是薄薄的一层,但如果把它放在显微镜下面仔细观察,就会清楚的看到皮肤由外及里共分为三层:皮肤的最外层叫表皮:中间一层叫真皮:最里面的一层叫皮下组织。表皮是皮肤的最外层包含各种大小不同的鳞片状上皮细胞,由基层发育而成。此基层位于真皮之上,在发育过程中不断的产生新细胞。表皮细胞还能够进一步细分为基底层、棘层、颗粒层、透明层和角质层。表皮细胞之间排列非常紧密,能够对一些化学物质和水分有屏障作用,因此能够保护真皮和皮下组织[1]。皮肤衰老的主要原因发生在真皮,真皮是皮肤的主要构成部分。真皮由弹力纤维、胶原纤维和网状纤维组成的结缔组织与纤维束间的无定型基质构成,真皮的厚度随身体部位功能的不同而异。皮肤衰老或者长期过度日晒会使皮肤产生皱纹、弹性松弛,是胶原纤维及弹力纤维变性或断裂的结果。
人体会在内源性或外源性因素的诱导下产生活性氧(ROS)和活性氨(RNS)。与此同时,人体自身防御系统能够清除细胞中的自由基。但是,随着年龄的增长和外界刺激如环境污染、紫外光照射和炎症条件下,自由基的产生会增加数倍[2]。自由基的产生和通过人体自身抗氧化系统的清除导致氧化应激[3],氧化应激会损伤很多生物分子,因而导致多种疾病的形成[4]。在这种情况下,通过外源性补充抗氧化剂来清除体内自由基就显得尤为重要[3]。在食品和化妆品领域,由于众多合成的抗氧化剂存在潜在的致癌性而让人们对天然的抗氧化剂的研究与应用越来越重视[6]。芳香植物,尤其是其挥发油是很好的天然抗氧化剂[7]。如今,人们致力于研究天然植物的挥发油,因为其相对安全、容易被消费者接受、在化妆品、食品、药品等领域应用广泛、容易获得并且相对廉价[8]。
紫外照射是黑色素形成的一一个关键因素[9],之前有研究证明了紫外照射诱导的ROS和RNS过量产生会使黑色素增多[10]。使用防晒霜是目前最流行的减少紫外线对皮肤损伤的方法[11]。纳米二氧化钛广泛应用于防晒产品中[12]。但是,最近有研究表明纳米二氧化钛促进含水环境中颗粒表面光诱导产生活性氧[13,14]。另一方面,在正常的阳光照射环境下,汗水的蒸发能够提高皮肤表面亚硝酸盐的量[15-17]。因此,由于紫外光的照射,纳米二氧化钛和亚硝酸盐会导致蛋白质氧化和酪氨酸硝化。蛋白质酪氨酸硝化不仅会引起蛋白质结构和功能的改变,而且会额外增加光引起的氧化损伤。因此,蛋白质氧化和酪氨酸硝化可以认为是纳米二氧化钛对皮肤的损伤。
本章通过三种不同抗氧化试验来研究三种挥发油的抗氧化活性,同时研究三种挥发油对UV-TiO2-NO2 诱导的蛋白质氧化和硝化的保护作用。
4.2材料与方法
4.2.1试剂与仪器
4.2.1.1主要试剂
2,4-二硝基苯肼(DNPH).牛血清蛋白( BSA)、二叔丁基对甲酚( Butylated hydroxytoluene, BHT)、兔多克隆抗二硝基苯酚抗体、兔多克隆抗3-硝基酪氨酸抗体:美国Sigma 公司;超氧阴离子自由基试剂盒:南京建成生物研究所;三羟甲基氨基甲烷(Tris)、2,4-二硝基苯肼( 2,4-dinitrophenylhydrazine, DNPH)、考马斯亮蓝R-250、十二烷基硫酸钠(SDS)、 吐温(Tween-20)、 考马斯亮蓝G-250、丽春红:美国Amersco公司;ECL发光试剂、辣根过氧化物酶标记的羊抗兔IG(H+L): 美国Pierce 公司;甲叉双丙烯酰胺、溴酚蓝、丙烯酰胺:瑞典Pharmacia公司;硝酸纤维素膜:美国Millipore 公司。
4.2.1.2主要仪器
SPECTR Amax PULS384型酶标仪:美国Sunnyvale公司;
DYCP-40C型电泳槽:北京六一-仪器厂;
Milli-Q Synthesis超纯水系统:国美Millipore 公司;
SSQ-H型不锈钢恒温水浴锅:上海三 申医疗器械有限公司;
DYY-7C型电泳仪:北京六一仪器厂;
UV7500型紫外-可见分光光度计:.上海 天美科学仪器有限公司;
X型胶片及感光暗盒:美国Kodak 公司;
4.2.1.3溶液的配置:
SDS-聚丙烯酰胺凝胶溶液的配制
电泳工作液:
①30%(w/v)丙烯酰胺单体储存液: 29.2 g丙烯酰胺,0.8 g双丙烯酰胺,溶于100 mL二次水中,储藏于棕色瓶中4C保存备用;
②1.5 M Tris-HCl (pH= >8.8):将36.34g Tris碱溶于150 mL二次水中,用浓盐酸调节pH值至8.8, 最后加二次水到总体积为200 mL,4C保存;
③1.0 M Tris-HCl (pH=6.8): 12.10g Tris 碱溶于50 mL二次水中,用浓盐酸调节pH值至6.8,最后加二次水到总体积为100mL,4C保存;
④10%过硫酸胺:使用小管分装,于-20°C 保存;
⑤5x电泳缓冲液: 15.1 gTris碱,94g甘氨酸,5g SDS 溶于1000 mL二次水中,室温条件下保存;
1x样品缓冲液(10 mL): 1.0mL甘油,1.25 mL Tris-HCl(0.5M,pH=6.8), 1.5mL 20% SDS(w/v), 0.5mLβ_巯基乙醇,2.0mL 0.1%溴酚蓝,3.75 mL二次水,4'C保存;
考马斯亮蓝染色液: 0.25g 考马斯亮蓝R-250, 112.5mL 甲醇,25 mL冰醋酸,然后加二次水定容至总体积1000 mL;
考马斯亮蓝脱色液:甲醇50 mL,冰醋酸50 mL,二次水400 mL。
Western blot所用溶液的配制:
半干转移缓冲液: 5.8g Tris碱,2.9g 甘氨酸,200 mL甲醇,0.37 gSDS,然后加二次水至总体积1000 mL;
TBS-T:8.77g氯化钠,3.15 g Tris-HCl溶于950 mL二次水中,然后用1M氢氧化钠溶液调节pH值至7.4, 最后加入0.5 mL Tween-20并补加二次水至总体积为1000mL, 4C保存;
封闭液: 5%脱脂奶粉( TBS-T溶解);
丽春红染色液: 3%三氯乙酸(w/v),0.2% 丽春红(w/v),3%磺基水杨酸(w/v);
4.2.2超氧阴离子清除实验
运用超氧阴离子自由基试剂盒检测样品对超氧阴离子自由基的清除能力。溶液的配置和样品的检测方法按照试剂盒说明书操作。槲皮素作为阳性对照。超氧阴离子清除率的计算方法:
抑制率%= (Ablamk -Asample)/AblankX 100 Aplamk和Asample 分别表示不添加实验样品和添加实验样品时的吸光度。
4.2.3 ABTS阳离子(ABTS*)清除实验
ABTS阳离子清除实验参照Re等[18] 和Gao等[19的方法。ABTS 溶解于蒸馏水,浓度为7mM。溶液中加入MnO2粉末并在黑暗环境下室温保存超过24小时,用注射器式过滤器过滤并在黑暗环境中放置6小时后使用。获得的ABTS 阳离子以这种形式在黑暗环境中室温保存超过2天。含有ABTS阳离子的母液用乙醇稀释至其吸光度值在734 nm波长下为0.70 (+0.02)。每种样品稀释至适当的浓度。100μL稀释的样品与1.2mL稀释的ABTS阳离子溶液反应,从反应开始到2分钟,记录分光光度计在734nm波长处的吸光度值。
自由基清除率% = (Ablank -Asample)AblankXx 100
其中Ablank代表没有添加实验样品时的吸光度值,Asample代表添加实验样品时的吸光度值。槲皮素作为阳性对照。
4.2.4脂质过氧化的测定
实验方法参照Mihara and Uchiyamal20]和Hermes-Lima等[21。 亚油酸作为抗脂质过氧化的底物。将0.2804g 的亚油酸、0.2840g 的吐温20、 50 mL磷酸缓冲溶液(pH7.0)混合为均匀的乳浊液。乳浊液现用现配,不可存储。200 μL样品溶解于DMSO,加入2mL亚油酸乳状液,然后加入氯化亚铁(100 μL,0.6 mmol/L, 溶剂为3.5%的盐酸)和过氧化氢(400 μL,25 mmol/L);对照组为2500 μL磷酸缓冲溶液和200 uL样品溶液。剧烈震荡后,反应混合液在37°C 静置60分钟;加入2.0mL 1%的TAB溶液(含有0.002% BHT, 0.8% TCA, 0.8% SDS)在95°C反应30分钟。过氧化值通过分光光度计在550nm波长处的吸光度值来体现。Trolox作为阳性对照。
脂质过氧化的抑制率% = (Ablank-Asample)/ AblankX 100
其中Ablank表示未添加实验样品的吸光度值,Asample表示添加了实验样品的吸光度值。
4.2.5纳米TiO2- NO2 -UV诱导的蛋白质氧化和酪氨酸硝化测定
实验方法参照本实验室之前的方法[21并做略微修改。BSA(1.0mg/mL),NaNO2 (1.0 mmol/L)和悬浮的TiO2(50μg/mL) 与pH7.0的0.1 mol/L PBS充分混合(总体积3 mL)。混合液移入24孔板直接暴露在光源下。365 nm波长的紫外光照射时反应开始。365 nm波长紫外光照射4小时,10000 转/分钟离心15 分钟去除TiO2,上清液备用。
4.2.5.1蛋白质氧化测定
取处理好的.上清液100 μL,加入100μL含3%SDS的10 mmol/L DNPH,室温下反应45分钟(每隔几分钟使用漩涡仪充分混匀),冰浴下加入200 μL的中和液(2 mol/L Tris, 30%甘油)终止反应。然后样品与280μL的2x.上样缓冲液,混匀后于-20 C储存备用。进行10% SDS-PAGE。电泳完毕后,运用半干转移法将蛋白质转移到硝酸纤维素膜上,在室温条件下将印迹膜放入封闭液(5%脱脂奶粉)中搅动孵育1.5 小时。孵育完毕后,用TBS-T漂洗4次,每次10分钟。然后用兔多克隆抗3-DNP抗体(1: 4000 )孵育印迹膜(4 C,1.5 小时),接着继续用TBS-T漂洗4次,每次10分钟;然后室温下用辣根过氧化物酶标记的羊抗兔IgG (二抗,1: 4000) 孵育印迹膜1小时,再用TBS-T溶液漂洗5次,每次10分钟。最后采用化学发光法检测。
4.2.5.2蛋白质硝化的测定
150 μL.上清液与150 ul 2x.上样缓冲液混匀后于100 °C沸水中反应3分钟,进行10% SDS-PAGE。
SDS-聚丙烯酞胺凝胶电泳:在12% SDS -聚丙烯酰胺凝胶电泳中进行分离。
蛋白印迹:电泳完成以后,利用半干转移法将蛋白质转移到硝酸纤维素膜上,在室温条件下将印迹膜放入封闭液(5%脱脂奶粉)中孵育1.5 小时。孵育完毕后, TBS-T溶液漂洗4次,每次10分钟。然后用兔多克隆抗3-硝基酪氨酸抗体(一抗,1:800) 室温条件下孵育印迹膜1.5 小时,TBS-T溶液漂洗4次,每次10分钟;然后用辣根过氧化物酶标记的羊抗兔IgG( 二抗,1:3000) 室温条件下孵育印迹膜1小时,然后再用TBS-T溶液漂洗4次,每次10分钟。最后用化学发光法检测。
4.2.6数据统计
所有实验结果均至少重复3次,实验结果以X士SD来表示,差异显著性分析采用组间t检验法。p<0.05, 认为两组数据间有显著差异; p<0.01, 认为两组数据间有非常显著性差异。
4.3试验结果
4.3.1山苍子油及其活性成分对ABTS*的清除能力测定
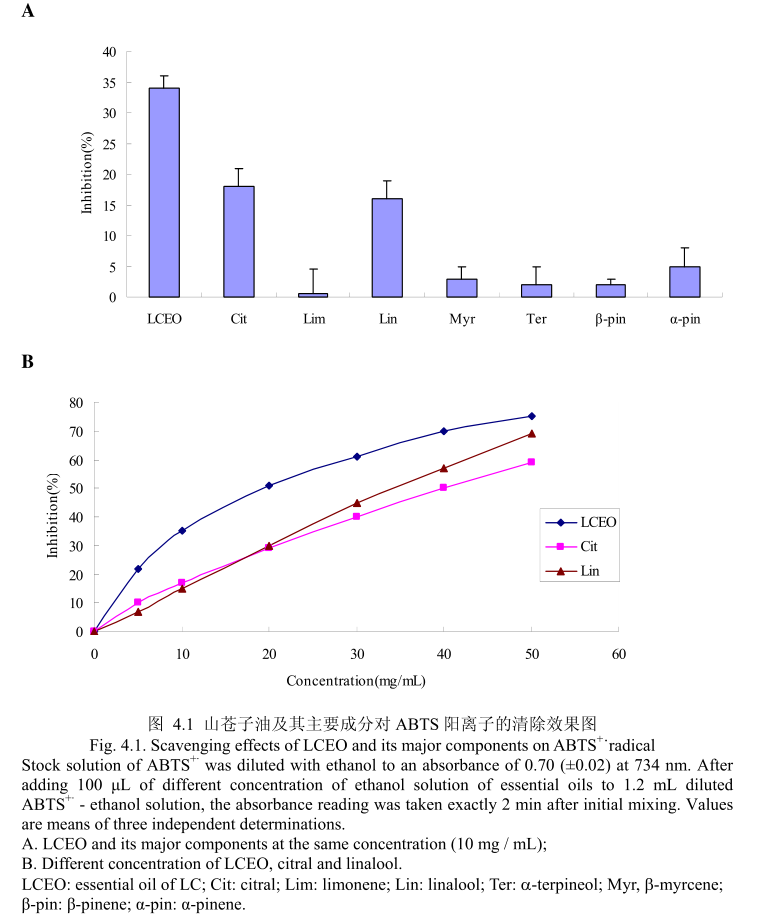
ROS在调节角蛋白细胞和黑素细胞增殖方面有重要作用,同时在调节黑素细胞生成黑色素方面也有重要作用。而能够清除ROS和抑制ROS产生的物质如抗氧化剂,能够减少色素沉着或者阻止紫外线诱导的黑素生成[23,24]。为了测定LCEO和其主要成分是否具有抗氧化活性,通过清除ABTS+来测定。LCEO和其主要成分对ABTS+的清除能力如图4.1 (A)。 LCEO在浓度为10mg/mL时被证明具有很强的抑制ABTS+产生的能力,柠檬醛和芳樟醇在相同浓度下表现出一定的抑制能力,柠檬烯、β-月桂烯、a-松油醇、β-蒎烯、a-蒎烯在该浓度下表现出的抑制活性可以忽略不计。结果表明柠檬醛和芳樟醇是LCEO中清除ABTS+的活性成分。如图4.1 (B),进一步测定了不同浓度下的LCEO和其活性成分对ABTS+的清除能力。从图中可以看出,ABTS+的清除能力与使用剂量相关。各种成分的ICso 的值按照逐渐递减的顺序为:柠檬醛>芳樟醇>
LCEO。从而证明LCEO比另外两种活性成分有着更强的清除ABTS+ 的能力。 将LCEO对ABTS+ 的清除能力与槲皮素比较,LCEO的半抑制浓度为17.75+0.89mg/ mL,低于槲皮素的半抑制浓度为4.5+0.9 ug / mL。
4.3.2 山苍子油及其活性成分对 0-·的清除能力测定
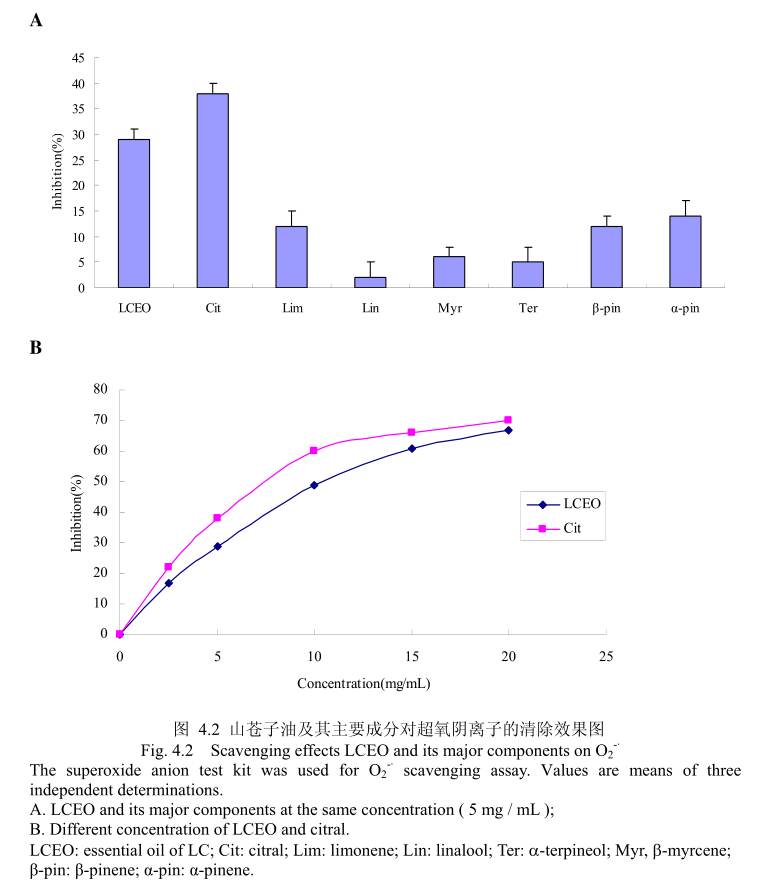
山苍子油及其活性成分对〇的清除能力测定如图4.2(A)所示,在浓度为5mg/mL 的情况下,柠檬醛和LCEO对O的抑制率分别为37%和 28%,而其它组分在相同的浓度下表现出的抑制能力在5%-15%之间。证明LCEO 具有一定的清除 O,的能力,其主要活性成分为柠檬醛。如图4.2(B)所示,当LCEO 和柠檬醛的浓度从 2.5mg/mL增加到20mg/mL 时,LCEO 和柠檬醛对 O的清除活性增加。而且,LCEO 和柠醛表现出相似的清除能力。将LCEO的抗氧化能力与槲皮素作比较,LCEO对0的半抑制浓度为10.2+0.12mg/m,而皮素的半抑制浓度为 10.3+0.4μg/m。结果表明皮素的抗氧化能力强于 LCEO。
4.3.3 LCEO 和柠檬醛对 UV-TiO,-NO,催化的蛋白质氧化损伤的保护
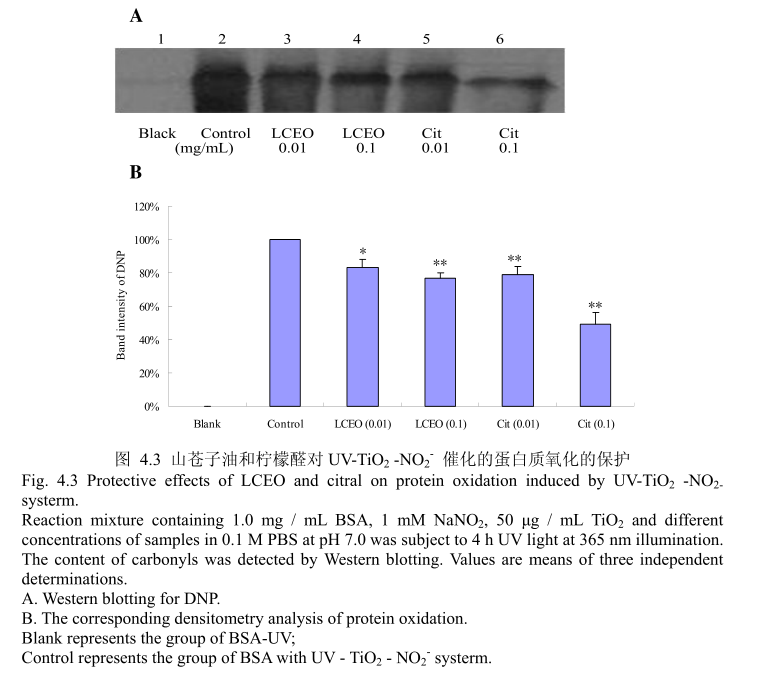
蛋白质氧化是对蛋白质的氧化修饰,改变了蛋白质的结构和功能。蛋白质的氧化常常存在于生理和病理状态下。因此,蛋白质氧化可能存在于纳米TO,对皮肤的毒性过程中。实验使用牛血清蛋白(BSA)作为模型蛋白,然后用SDS-PAGE和蛋白质印迹法检测。如图4.3(A)和(B),蛋白质氧化通过添加0.01mg/mL(最低浓度)的LCEO或柠檬醛被抑制。与此同时,所有的样品都具有较强的保护作用,并且明显呈剂量相关性。当LCEO或柠醛的浓度达到0.1mg/m时,表现出的抑制蛋白质氧化的能力更强。此外,在相同的浓度条件下,柠檬醛比LCEO 表现出更强的抑制蛋白质氧化的能力。
4.3.4LCEO 和柠檬醛对 UV-TiOz-NO,催化的蛋白质硝化损伤的保护
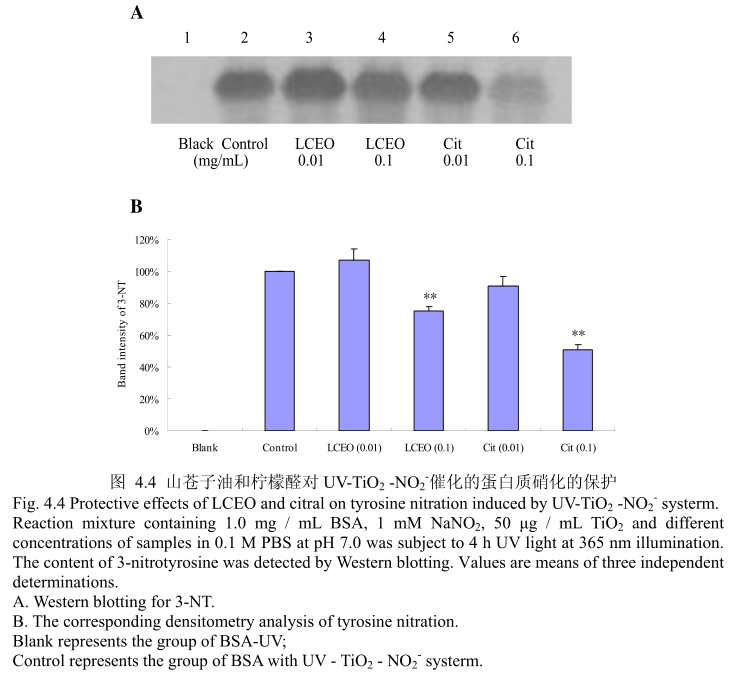
蛋白质硝化是机体在氮氧化物存在的条件下氧化损伤的特殊形式,主要是指蛋白质中含有的酪氨酸残基被硝化成3-硝基酪氨酸(3-Nitro-Tytosine, 3~NT),即一个硝基(-NO2)添加在酪氨酸残基的羟基邻位上。蛋白质硝化在病理状态下常伴随着活性氮和活性氧的增加而产生。目前,蛋白质酪氨酸硝化已经被确认为是一-种重要的蛋白质翻译后的修饰,并且与炎症和神经退行性疾病、心血管疾病等多种病症密切相关,其产物3-硝基酪氨酸(3-NT) 作为NO、ONOO~等活性氮的衍生物,可以被认为是许多生理和病理状态下的生物标志物(Biomarker)[5]126。将3-硝基酪氨酸作为酪氨酸硝化的检测指标,LCEO和柠檬醛对蛋白质硝化的抑制效果如图4.4 (A)和(B)。当LCEO或柠檬醛的的浓度为0.01 mg/mL时,并没有表现出较强的抑制蛋白质硝化的能力,当LCEO 或柠檬醛的浓度达到0.1 mg/ mL时,才能表现出较强的蛋白质硝化的抑制活性。
4.3.5丁香花蕾油及其主要成分对O2的清除能力测定
超氧阴离子作为更多活性氧的前体对细胞结构有着很大的损伤,导致组织损伤和不同的疾病[26]。
丁香花蕾油和其主要成分丁香酚对O2” 的清除活性如图4.5。结果表明丁香花蕾油的比丁香酚表现出更强的清除O2”的能力。丁香花蕾油的浓度分别为10ug/mL、30 ug/mL、60μg/mL时,超氧阴离子的清除能力为29.8%, 41.5%,50.5%,而对于相同浓度的丁香酚,其对超氧阴离子的清除能力分别为20.4%, 25.3%,30.8%。当丁香酚或丁香花蕾油的量增加时,抑制能力并没有明显的变化。这可能是由于其在水中的溶解性较低的缘故,当溶解到一定量时,增加的量并不能溶解于测试溶剂,从而不能发挥抑制作用。丁香花蕾油的对超氧阴离子的半抑制浓度为58ug/mL,低于槲皮素的半抑制浓度为10.3 ug / mL。
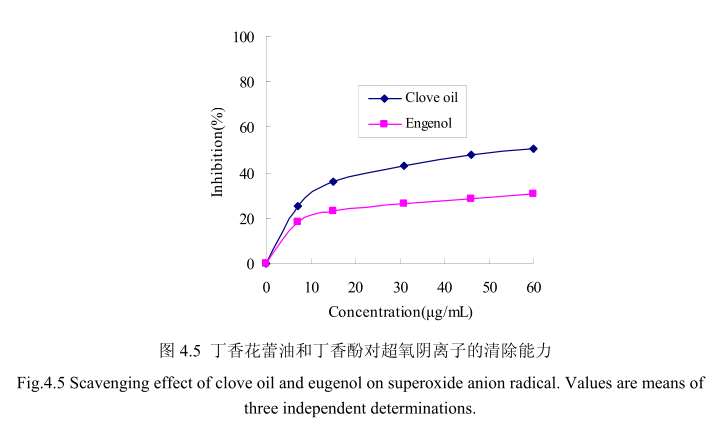
4.3.6 丁香花蕾油及其主要成分对 ABTS*的清除能力测定
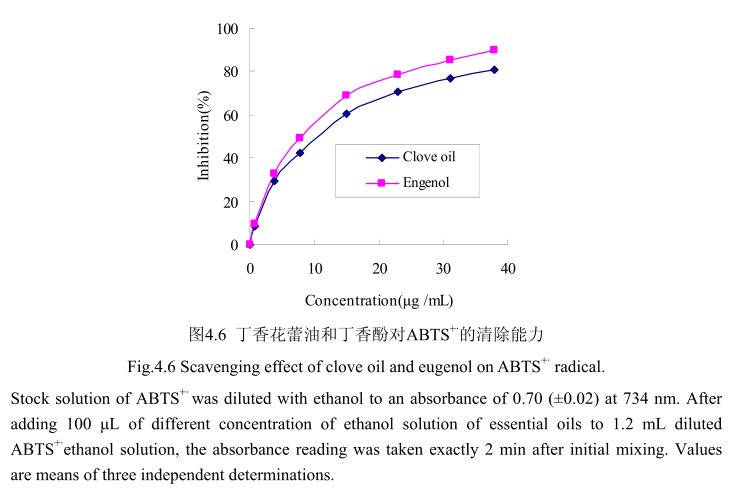
丁香花蕾油和其主要成分丁香酚对ABTS+的清除活性如图4.6 所示。丁香花蕾油和丁香酚都表现出较高的清除活性。当丁香花蕾油的浓度为0.8-38μg/mL时,其清除活性为8.7%-80.9%, 在相同浓度范围内,丁香酚的清除率为9.7%-89.6%。当丁香花蕾油的浓度从0μg/mL逐渐增加到15μg/mL时,对ABTS+的清除率急剧增加(从0增加到70%左右);当丁香酚的浓度从0μg/mL逐渐增加到15μg/ mL时,对ABTS+的清除率亦明显增加(从0增加到60%左右)。当丁香花蕾油的浓度从15 ug/mL逐渐增加到40 ug/mL时,对ABTS+的清除率增加量逐渐变缓(从70%增加到89.6%);当J香酚的浓度从15 ug/ mL逐渐增加到40μg/mL时,对ABTS+的清除率从60%增加到79%。丁香花蕾油和丁香酚的半抑制率分别为8.5 μug/ mL和7.7 ug/ mL,与槲皮素的半抑制浓度4.5 ug/ mL相比差别不大。
4.3.7香茅油及其主要成分对O2-.的清除能力测定
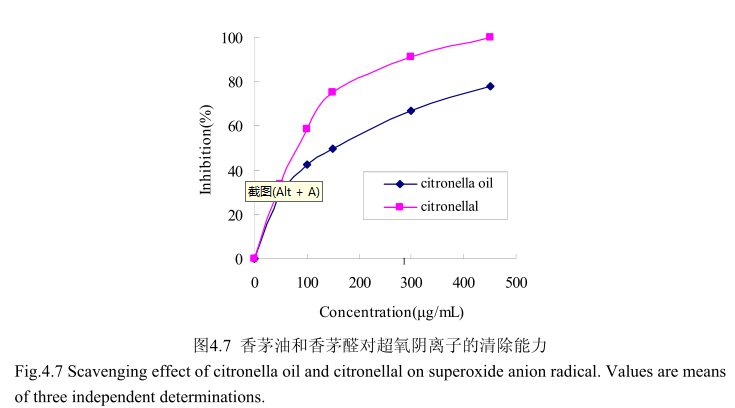
香茅油及其主要成分香茅醛对超氧阴离子的清除活性如图4.7所示。香茅油的半抑制浓度(ICso:150μg/mL)大于香茅醛的半抑制浓度(ICso:90μg/mL),说明香茅油对超氧阴离子的抑制活性弱于香茅醛。当香茅醛的浓度为450mg/m时,其对超氧阴离子的清除活性达到100%,而香茅油在该浓度下的清除活性仅为78%。香茅油对超氧阴离子的半抑制浓度为150mg/mL,说明其对超氧阴离子的清除能力弱于槲皮素(10.3μg/mL)。当香茅油的浓度从0g/m增加到50μg/m时,对超氧阴离子的抑制能力显著增加,随着香茅油的浓度逐渐增大,对超氧阴离子的清除能力的增加量逐渐变缓。当香茅醛的浓度0mg/m增加到150mg/m时,对超氧阴离子的清除能力显著增加(从0增加到78%左右),而当香茅醛的浓度从150g/m增加到500μg/m 时,清除能力增加的较为缓慢(从78%增加到 100%)。
4.3.8 香茅油及其主要成分对 ABTS*的清除能力测定
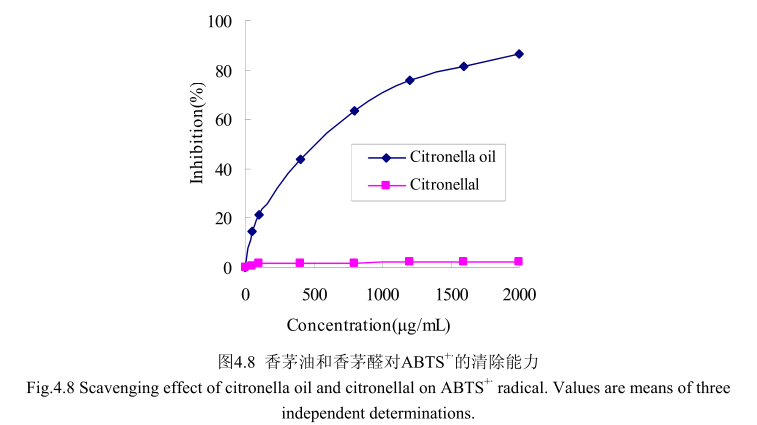
如图4.8所示,当香茅油的浓度范围在50-2000g/m时,其对 ABTS”的清除率为 14.5%-86.4%。尽管香茅油在低浓度时100g/m 时的抑制率仅为20%,但是香茅油的浓度从0g/m增加到100gm时,对ABTS*的清除能力显著增加;随着香茅油的浓度从100g/mL增加到2000g/mL时,对ABTS*的清除能力缓慢增加,且增加趋势逐渐变缓。而香茅油的主要成分香茅醛在相同的浓度条件下并没有表现出清除能力。而香茅油半抑制浓度为500g/m,远远大于皮素 4.5μg/m,从而说明香茅油的抗氧化活性较弱,
4.3.9丁香花蕾油和香茅油对脂质过氧化的保护作用
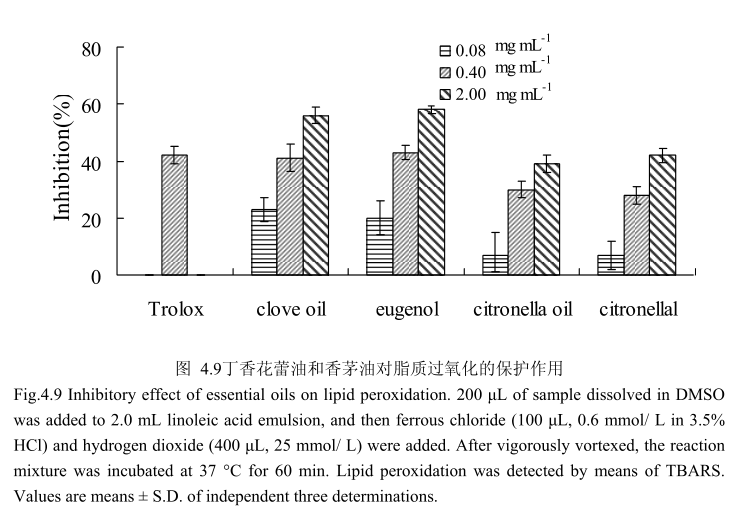
丁香花蕾油和香茅油及其主要成分对脂质过氧化的抑制作用如图4.9所示,丁香花蕾油、香茅油、丁香酚、香茅醛都具有抗脂质过氧化的作用,并且随着剂量的增加,抗氧化活性逐渐增强。在相同的浓度条件下,丁香花蕾油和丁香酚的抗氧化能力强于香茅油和香茅醛。当浓度为0.4mg/m时,丁香花蕾油和丁香酚抗脂质过氧化的能力与 Trolox(阳性对照)相似。
4.3.10 丁香花蕾油和香茅油对 UV-TiO,-NO,催化的蛋白质氧化损伤的保护
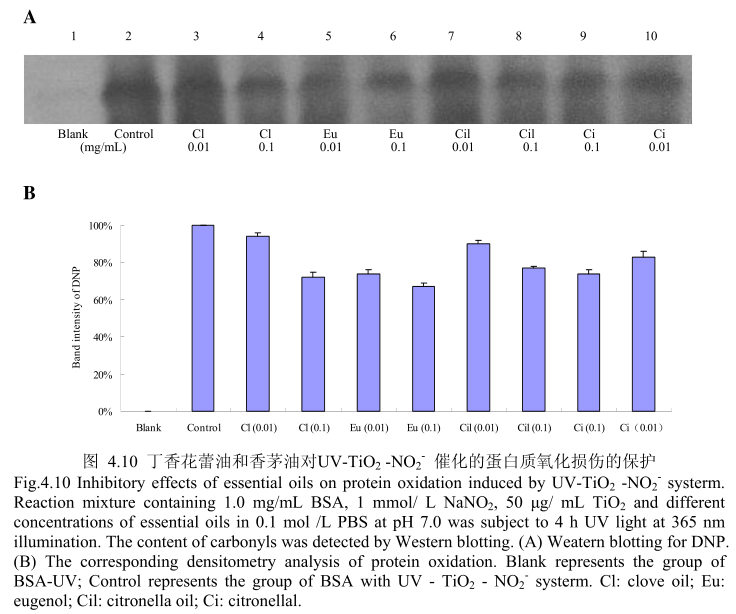
丁香花蕾油和香茅油对 UV-TiO2 -NO,催化的蛋白质氧化的抑制作用如图4.10A 和 B所示,当丁香花蕾油、丁香酚、香茅油、香茅醛以最低浓度0.01mg/ml 进行加样时,都具有抑制蛋白质氧化的能力。在该浓度下,丁香花蕾油和香茅油表现出相似的抑制蛋白质氧化的能力,丁香酚的抑制蛋白质氧化的能力强于丁香花蕾油,香茅醛的抑制蛋白质氧化的能力强于香茅油,丁香酚的抑制蛋白质氧化的能力强于香茅醛。当加样浓度增加到0.1mg/m 时,丁香花蕾油、丁香酚、香茅油、香茅醛均具有较好的抗氧化能力。在该浓度下,丁香花蕾油、香茅油、香茅醛表现出相似的抑制蛋白质氧化的能力,但在该浓度下其抗氧化活性弱于丁香酚。
4.3.11 丁香花蕾油和香茅油对 UV-TiO,-NO,催化的蛋白质硝化损伤的保护
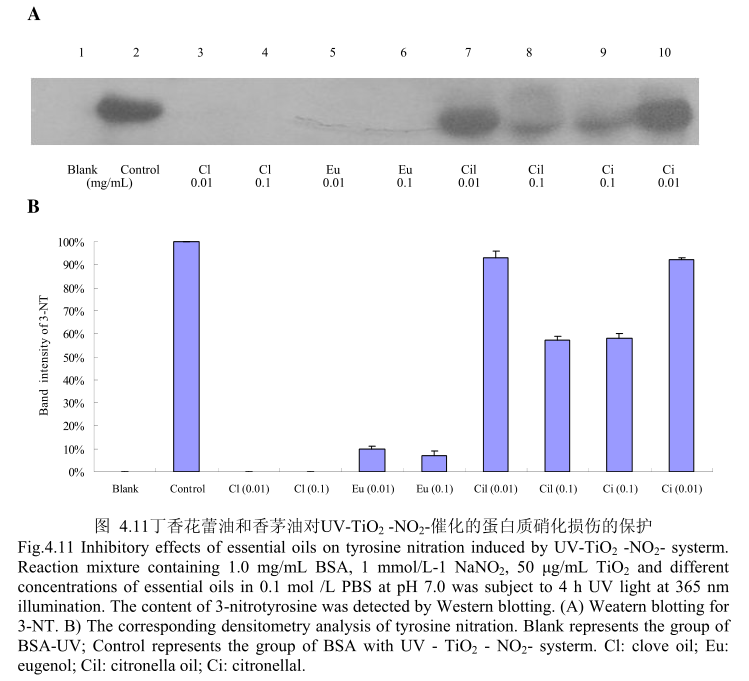
丁香花蕾油和香茅油对UV-TiO2-NO,催化的蛋白质硝化的抑制作用如图4.11,当丁香花蕾油的浓度为0.01mg/m和0.1mg/m 时,均能够完全抑制硝化的产生。而香茅油和其主要成分香茅醛在浓度为0.01mg/m时,几乎没有抑制蛋白质硝化的能力,当香茅油和香茅醛的浓度为0.1mg/mL时,表现出一定的抑制蛋白质硝化的能力。
4.4讨论
挥发油是芳香植物的次生代谢产物,有着明显的气味,是具有挥发性的混合物。在自然界,挥发油在保护植物方面有着重要作用,如抗病毒、抗细菌、杀灭昆虫和减少昆虫食用植物的欲望。目前已经发现大约3000 种挥发油,而其中有300种具有重要的商业价值,被用于制药、农业、食品、卫生、化妆品和香料领域。很多挥发油同时也是天然的抗氧化剂,如薄荷油[27-29]、迷迭香油[30-33]、鼠尾草油[34]、芫荽子油[27,29]、小茴香油[35]、百里香油[36]等。挥发油能够作为香精的原料加入到化妆品中,同时也可以作为天然防腐剂或者活性成分用于化妆中。此外,挥发油常常与其它植物油脂按照适当的比例调配后直接涂抹于皮肤,不仅能够放松神经,还能发挥挥发油特有的功效。因此,研究出一-些具有特殊功效的挥发油具有重要意义。
皮肤是人体抵御损伤的屏障,是将人体与外界环境隔开的重要屏障。持续的暴露在紫外光下会导致皮肤损伤相关的众多并发症,当紫外光引起的损伤超过人体黑.色素提供的保护能力时就会形成晒斑[37,38]。系外线诱导皮肤光损伤,导致皮肤的细胞基质发生改变,引起皱纹、皮肤松弛、粗糙、色素沉着、表皮增厚和结缔组织变化[39,40]。结缔组织各组分( 包括胶原、蛋白聚糖类、糖蛋白类)之间的平衡被打破
会导致不利影响,如表皮成纤维细胞光老化。皮肤自身具有抗氧化防御能力,能够清除包含自由基在内的活性氧,但是如果紫外光达到一定的强度时会超出这些防御系统的清除能力,从而导致自由基损伤细胞的组分,如蛋白质、脂类和DNA[41.42]。活性氧诱导的氧化应激能够最终导致细胞凋亡或细胞坏死[43]。活性氧在人体内的积累,在人体皮肤内源性损伤和光老化方面起着决定性作用,同时与皮肤炎症和皮肤癌有着密切的关系[44,45]。本章通过体外实验研究了丁香花蕾油、山苍子油、香茅油对自由基的清除能力。通过实验发现,LCEO在浓度为10 mg 1 mL时被证明具有很强的抑制ABTS阳离子产生的能力,柠檬醛和芳樟醇在相同浓度下表现出一定的抑制能力,柠檬烯、β-月桂烯、a-松油醇、β-蒎烯、a-蒎烯在该浓度下表现出的抑制活性可以忽略不计。LCEO对ABTS阳离子的半抑制浓度为17.75+0.89mg/ mL。在浓度为5mg/mL 的情况下,柠檬醛和LCEO对O2~ 的抑制率分别为37%和28%,而其他组分在相同的浓度下表现出的抑制能力在5%-15%之间。
LCEO对02”的半抑制浓度为10.2+0.12mg1mL。当香茅油的浓度范围在50-2000 μg / mL时,其对ABTS+的清除率为14.5%-86.4%。 从而说明香茅油对ABTS+的清除能力较弱。香茅油对超氧阴离子的半抑制浓度为150 ug/mL。丁香花蕾油和丁香酚都表现出较高的清除活性。当丁香花蕾油的浓度为0.8-38μg/mL时,其清除活性为8.7%-80.9%, 在相同浓度范围内,丁香酚的清除率为9.7%-89.6%。丁香花蕾油和丁香酚的半抑制率分别为8.5 μg/mL和7.7 μg/ mL。丁香花蕾油的浓度为10 ug/mL、30 μg/mL、60 μg/ mL时,超氧阴离子的清除能力为29.8%, 41.5%, 50.5%, 而对于相同浓度的丁香酚,其对超氧阴离子的清除能力分别为20.4%, 25.3%, 30.8%。丁香花蕾油的对超氧阴离子的半抑制浓度为58 ug. / mL。三种挥发油中丁香花蕾油对ABTS阳离子的清除能力最强,而其主要成分丁香酚对ABTS阳离子的清除能力更强,从而说明丁香花蕾油及其主要成分丁香酚有较强的抗氧化能力,与相关文献报道一致[46,47]。 而山苍子油对超氧阴离子的清除能力最强,说明山苍子油也具有较强的抗氧化能力。在脂质过氧化的抑制作用方面,丁香花蕾油、香茅油、丁香酚、香茅醛都具有抗脂质过氧化的作用,并且随着剂量的增加,抗氧化活性逐渐增强。丁香花蕾油和丁香酚的抗氧化能力强于香茅油和香茅醛。而山苍子油几乎没有抗脂质过氧化的能力。
蛋白质硝化是蛋白质氧化的一一种特殊形式,两者都是非常重要的蛋白质翻译后修饰,它们对蛋白质的功能产生非常重要的影响。而纳米二氧化钛在防晒产品中的广泛应用,紫外线的照射和皮肤表面汗液蒸发产生的亚硝酸盐为蛋白质的氧化和酪氨酸的硝化提供了充足的条件。本章研究了山苍子油、香茅油、丁香花蕾油对UVTiO2 - NO2催化的蛋白质硝化和蛋白质氧化的抑制作用,发现当LCEO或柠檬醛的浓度为0.01mg/mL时能够抑制蛋白质氧化,柠檬醛的抑制蛋白质氧化的能力强于LCEO,并且抑制能力呈剂量相关性。而对于抑制蛋白质的硝化,仅当LCEO和柠檬醛的浓度达到0.1 mg/ mL时才表现出较强的抑制蛋白质硝化的能力。当丁香花蕾油、丁香酚、香茅油、香茅醛以最低浓度0.01 mg / mL进行加样时,都具有抑制蛋白质氧化的能力。当加样浓度增加到0.1 mg/mL时,每种样品均具有较好的抗氧化能力。丁香花蕾油、香茅油、香茅醛表现出相似的保护作用,但在相同浓度下其抗氧化活性弱于丁香酚。对于蛋白质硝化的抑制作用方面,当丁香花蕾油和丁香酚的浓度为0.01mg/m时,能够完全抑制硝化的产生。而香茅油和其主要成分香茅醛仅在高浓度0.1mg/m 时,表现出一定的抑制蛋白质硝化的能力。
4.5 本章小结
在本章中,通过山苍子油、香茅油、丁香花蕾油体外清除ABTS和超氧阴离子实验,研究三种精油的抗氧化能力。我们发现丁香花蕾油及其主要成分丁香酚具有较强的清除ABTS*的能力,山苍子油和其主要成分柠檬醛具有较强的清除超氧阴离子的能力。丁香花蕾油、香茅油、丁香酚、香茅醛都具有抗脂质过氧化的作用,并且随着剂量的增加,抗氧化活性逐渐增强。丁香花蕾油和丁香酚的抗氧化能力强于香茅油和香茅醛。而山苍子油几乎没有抗脂质过氧化的能力。丁香花蕾油、丁香酚、香茅油、香茅醛、山苍子油、柠檬醛均具有较强的抑制蛋白质氧化的能力,其中丁香酚的抑制蛋白质氧化的能力最强。丁香花蕾油和丁香酚具有很强的抑制酪氨酸硝化的能力,山苍子油、柠檬醛、香茅油、香茅醛仅在较高浓度时表现出较强的抑制蛋白质硝化的能力。
4.6参考文献
[1] 涂国荣,张利兴,苏昆.自由基与皮肤衰老的关系及天然抗衰老植物在化妆品中的应用,日用化学品科学,1999,8:82-85
[2] Singh HP, Mittal S, Kaur S, et al. Characterization and antioxidant activity of essential oils from fresh and decaying leaves of Eucalyptus tereticornis. Journal of Agricultural and Food Chemistry, 2009, 57(15):6962-6966
[3] Edris AE. Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: a review. Phytotherapy Research, 2007, 21(4):308-323
[4]LuN,LiX,Li
J. Ntrative and oxidative modifications of enolase are
associated with iron in iron-overload rats and in vitro. Journal of Biological
Inorganic Chemistry, 2011, 16(3):481-490
[5] Aidi Wannes W, Mhamdi B, Sriti J, et al. Antioxidant activities of the essential
oils and methanol extracts from myrtle (Myrtus communis var. italica L.) leaf,
stem and flower. Food and Chemical Toxicology, 2010, 48(5):1362-1370
[6] Kilani S, Ledauphin J, Bouhlel I, et al. Comparative study of Cyperus rotundus essential oil by a modified GC/MS analysis method. Evaluation of its antioxidant, cytotoxic, and apoptotic effects. Chemistry & Biodiversity, 2008, 5(5):729-742
[7] Mechergui K, Coelho JA, Serra MC, et al. Essential oils of Origanum vulgare L. subsp. glandulosum (Desf.) letswaart from Tunisia: chemical composition and antioxidant activity. Journal of the Science of Food and Agriculture, 2010, 90(10):1745-1749
[8] Sutthanont N, Choochote W, Tuetun B. Chemical composition and larvicidal activity of edible plant-derived essential oils against the pyrethroid-susceptible and -resistant strains of Aedes aegypit(Diptera:Culicidae). Journal of Vector Ecology, 2010, 35:106-115
[9] Panich U, Kongtaphan K, Onkoksoong T. Modulation of antioxidant defense by Alpinia galangal and curcuma aromatica extracts correlates with their inhibition of UVA-induced melanogenesis. Cell Biology Toxicology, 2010, 26:103-116
[10] Panich U, Tangsupa a-nan V, Onkoksoong T, et al. Inhibition of UV A -mediated melanogenesis by ascorbic acid through modulation of antioxidant defense and nitric oxide system. Archives of Pharmacal Research, 201 1, 34(5):811-820
[11] Burnett ME, Wang SQ. Current sunscreen controversies: a critical review.
Photodermatology, photoimmunology & photomedicine, 2011, 27(2):58-67
[12] Tyner K, Wokovich A, Godar D, et al. The state of nano-sized titanium dioxide
(TiO2) may affect sunscreen performance. International Journal of Cosmetic Science, 201 1,33(3):234-244
[13] Kiss B, Biro T, Czifra G, et al. Investigation of micronized titanium dioxide penetration in human skin xenografts and its effect on cellular functions of human skin-derived cells. Experimental dermatology, 2008, 17(8):659-667
[14] Shen B, Scaiano,English A. Zeolite encapsulation decreases TiO2-photosensitized ROS generation in cultured human skin fibroblastst. Photochemistry and Photobiology, 2006, 82(1): 5-12
[15] Paunel AN, Dejam A, Thelen S, et al. Enzyme-independent nitric oxide formation during UVA challenge of human skin: characterization, molecular sources, and mechanisms. Free Radical Biology and Medicine, 2005, 38(5):606-615
[16] Oplander C, Cortese M, Korth H. The impact of nitrite and antioxidants on ultraviolet-A-induced cell death of human skin fibroblasts. Free Radical Biology Medicine, 2007, 43(5):8 18-829
[17] Weller R, Pattullo S, Smith L, et al. Nitric oxide is generated on the skin surface by reduction of sweat nitrate. Journal of Investigative Dermatology, 1996, 107(3):327-331.
[18] Re R, Pellegrini N, Proteggente A, et al. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology Medicine, 1999, 26(9-10):1231-1237
[19] Gao Z, Xu H, Chen X, et al. Antioxidant status and mineral contents in tissues of rutin and baicalin fed rats. Life Sciences, 2003, 73( 12):1599- 1607
[20] Mihara M, Uchiyama M. Determination of malonaldehyde precursor in tissues by thio barbituric acid test. Analytical Biochemistry, 1978, 86(1):271-278
[21] Hermes-Lima M, Willmore WG, Storey K B. Quantification of lipid peroxidation in tissue extracts based on Fe (II) xylenol orange complex formation. Free Radical Biology and Medicine, 1995,19(3):271-280
[22] Lu N, Zhu Z, Zhao X, et al. Nano titanium dioxide photocatalytic protein tyrosine nitration: A potential hazard of TiO2 on skin. Biochemical and Biophysical
Research Communications, 2008, 370(4):675-680
[23] Sapkota K, Park SE, Kim JE, et al. Antioxidant and anti melanogenic properties of chestnut flower extract. Bioscience, Biotechnology, and Biochemistry, 2010,
74(8):1527-1533
[24] Khazaeli P, Goldoozian
R, Shariffar F. An evaluation of extracts of five
traditional medicinal plants from Iran on the inhibition of mushroom tyrosinase
activity and scavenging of free radicals. International Journal of Cosmetic
Science, 2009, 31(5):375-381
[25] Beal MF. Oxidatively modified proteins in aging and disease. Free Radical Biology and Medicine, 2002, 32(9):797-803
[26] Gutteridge J, Richmond R, Halliwell B. Oxygen free-radicals and lipid peroxidation: Inhibition by theprotein caeruloplasmin. FEBS letters, 1980, 112(1): 269-272
[27] Lagouri V, Blekas G, Tsimidou M, et al. Composition and antioxidant activity of essential oils from oregano plants grown wild in Greece. Zeit schrift fir Lebensmitteluntersuchung und Forschung A, 1993, 197(1):20-23
[28] Lagouri V, Boskou D. Screening for antioxidant activity of essential oils obtained from spices. Developments in Food Science, 1995, 37(1):869-879
[29] Baratta MT, Dorman HJD, Deans SG, et al. Chemical composition, antimicrobial and antioxidative activity of laurel, sage, rosemary, oregano and coriander essential oils. Journal of Essential Oil Research, 1998, 10(6):618-627
[30] Farag R, Badei AZMA, Hewedi F, et al. Antioxidant activity of some spice essential oils on linoleic acid oxidation in aqueous media. Journal of the American Oil Chemists' Society, 1989, 66(6):792-799
[31] Maestri DM, Zygadlo JA, Lamarque AL, et al. Effect of some essential oils on oxidative stability of peanut oil. Grasas y aceites, 1996, 47(6):397-400
[32] Dang M, Takacsova M, Nguyen D, et al. Antioxidant activity of essential oils from various spices. Food/Nahrung, 2001, 45(1):64-66
[33] Saricoban C,Ozcan M. Antioxidative activity of rosemary (Rosmarinus officinalis L.) and sage (Salvia fruticosa L.) essential oils in chicken fat. Journal of Essential Oil Bearing Plants, 2004, 7(1):91-95
[34] Tepe B, Daferera D, Sokmen A. Antimicrobial and antioxidant activities of the essential oil and various extracts of Salvia tomentosa Miller (Lamiaceae). Food Chemistry, 2005, 90(3):333- -340
[35] Farag R, Ali M, Taha S. Use of some essential oils as natural preservatives for butter. Journal of the American Oil Chemists Society, 1990, 67(3):188-191
[36] Jukic M, Milos M. Catalytic oxidation and antioxidant properties of thyme essential oils (Thymus vulgare L). Croatica Chemical Acta, 2005,78(1):105-110
[37] Agar NS, Halliday GM, Barnetson RSC, et al. The basal layer in human squamous tumors harbors more UVA than UVB fingerprint mutations: a role for UVA in human skin carcinogenesis. Proceedings of the National Academy of Sciences of the United States of America, 2004, 101(14):4954-4959
[38] Ryu BM, Qian ZJ, Kim MM, et al. Anti-photoaging activity and inhibition of matrix metalloproteinase (MMP) by marine red alga, Corallina pilulifera methanol extract. Radiation Physics and Chemistry, 2009, 78(2):98- 105
[39] Kondo S. The roles of cytokines in photoaging. Jourmal of Dermatological Science, 2000, 23(1):30-36
[40] Rittie L, Fisher GJ. UV-light-induced signal cascades and skin aging. Ageing Research Reviews, 2002, 1(4):705-720
[41] Kehrer JP. Free radicals as mediators of tissue injury and disease. Critical Reviews in Toxicology, 1993, 23(1):21-48
[42] Aruoma O. Nutrition and health aspects of free radicals and antioxidants. Food and Chemical Toxicology, 1994, 32(7):671-683
[43] Fiers W, Beyaert R, Declercq W, et al. More than one way to die: apoptosis, necrosis and reactive oxygen damage. Oncogene, 1999, 18(54):7719-7730
[44] Record IR, Dreosti IE, Konstantinopoulos M, et al. The influence of topical and systemic vitamin E on ultraviolet light-induced skin damage in Hairless Mice. Nutrition and Cancer, 1991,16(3):219-225
[45] Kawaguchi Y, Tanaka H, Okada T, et al. The effects of ultraviolet A and reactive oxygen species on the mRNA expression of 72-kDa type IV collagenase and its tissue inhibitor in cultured human dermal fibroblasts. Archives of Dermatological Research, 1996,288( 1):39-44
[46] Ogata M, Hoshi M, Urano S, et al. Antioxidant activity of eugenol and related monomeric and dimeric compounds. Chemical and Pharmaceutical Bulletin-tokyo, 2000, 48( 10): 1467-1469
[47] Guan W, LiS, Yan R, et al. Comparison of essential oils of clove buds extracted with supercritical carbon dioxide and other three traditional extraction methods. Food Chemistry, 2007, 101(4):1558-1564
5全文总结
挥发油是芳香植物的次生代谢产物,安全性高,生物活性强。本文首先优化了水.蒸气蒸馏法提取山苍子油、丁香花蕾油、香茅油的条件,并在优化条件下,提取了三种挥发油,然后用GC-MS法分析了三种挥发油的成分;探讨了山苍子油、丁香花蕾油、香茅油及其主要成分柠檬醛、柠檬烯、芳樟醇、月桂烯、松油醇、丁香酚、乙酸丁香酯、β -石竹烯、香茅醛、香叶醇、香茅醇与酪氨酸酶活性的关系,并研究了山苍子油的主要成分柠檬醛对酪氨酸酶的抑制类型;测定了山苍子油、丁香花蕾油、香茅油和其主要成分柠檬醛、丁 香酚、香茅醛对ABTS阳离子和超氧阴离子自由基的清除能力,研究了丁香花蕾油、香茅油和其主要成分柠檬醛、丁香酚、香茅醛对脂质过氧化的抑制作用;考查了山苍子油、丁香花蕾油、香茅油和其主要成分柠檬醛、丁香酚、香茅醛对UV - TiO2- NO2引起的蛋白质氧化和硝化抑制作用。
5.1本论文的主要研究成果
(1)运用水蒸气蒸馏法萃取的自然晾干的山苍子果实的挥发油呈淡黄色。GC-MS分析水蒸气蒸馏法提取的山苍子油鉴定出19种化学成分,占总油的92.4%。其主要成分柠檬醛的相对含量占57.4%, 柠檬烯的含量占8.1%, 芳樟醇的含量为5.2%,β- 月桂烯的相对含量为4.7%, a-松油醇的相对含量为3.9%,β- 蒎烯的相对含量为2.7%, a -蒎烯的相对含量为1.9%。 同样用GC-MS分析产于中国广东的丁香花蕾油,鉴定出10 种物质,占总油的94.8%。 主要成分丁香酚的相对含量为76.8%,乙酸丁香酯的相对含量为9.5%,β-石竹烯的相对含量为6.0%。GC_MS分析的香茅叶的挥发油鉴定出17 种成分,占总油的93.5%。 其中
主要成分香茅醛的相对含量为33.9% 、香叶醇的相对含量为18.1% 、香茅醇的相对含量为11.1%
(2)通过酪氨酸酶抑制试验来评价三种挥发油及其主要成分对酪氨酸酶的抑制效果。结果表明山苍子油和其主要成分柠檬醛对酪氨酸酶单酚酶和二酚酶活性有抑制能力,对酪氨酸酶二酚酶的抑制能力强于酪氨酸酶单酚酶,且动力学结果表明,柠檬醛对酪氨酸酶是非竞争性抑制作用。丁香花蕾油和丁香酚对酪氨酸酶单酚酶具有一定的抑制作用,但是对酪氨酸酶二酚酶没有表现出任何抑制作用。而香茅油及其主要成分对酪氨酸酶没有抑制作用。
(3)通过山苍子油、香茅油、丁香花蕾油体外清除ABTS阳离子和超氧阴离子实验,研究三种精油的抗氧化能力。发现丁香花蕾油及其主要成分丁香酚具有较强的清除ABTS阳离子的能力,山苍子油和其主要成分柠檬醛具有较强的清除超氧阴离子的能力。丁香花蕾油、香茅油、丁香酚、香茅醛都具有抗脂质过氧化的作用,并且随着剂量的增加,抗氧化活性逐渐增强。丁香花蕾油和丁香酚的抗氧化能力强于香茅油和香茅醛。而山苍子油几乎没有抗脂质过氧化的能力。丁香花蕾油、丁香酚、香茅油、香茅醛、山苍子油、柠檬醛均具有较强的抑制蛋白质氧化的能力,其中丁香酚的抑制蛋白质氧化的能力最强。丁香花蕾油和丁香酚具有很强的抑制酪氨酸硝化的能力,山苍子油、柠檬醛、香茅油、香茅醛仅在较高浓度时表现出较强的抑制蛋白质硝化的能力。
5.2本论文创新之处
(1)以挥发油这种具有多重生物活性的天然产物为研究对象,结合GC-MS分析结果,从整体上研究挥发油的生物活性,然后深入研究挥发油中起主要的作用的化学成分,从而全面探讨挥发油的活性机理。
(2)同时探讨挥发油对酪氨酸酶的抑制作用和抗氧化作用。发现山苍子油及其主要成分柠檬醛具有较强的酪氨酸酶抑制活性和清除自由基的能力,说明山苍子油在化妆品领域具有皮肤美白和预防皮肤衰老的作用,在食品领域具有防止食品褐变和氧化变质的作用。
(3)选择UV- TiO2- NO2体系作为引起蛋白质氧化和硝化的模型,而这种模型是最为接近因使用化妆品导致的皮肤中蛋白质发生氧化和硝化的模型。通过实验发现,丁香花蕾油、丁香酚、香茅油、香茅醛、山苍子油、柠檬醛均具有较强的抑制蛋白质氧化的能力,其中丁香酚的抑制蛋白质氧化的能力最强。丁香花蕾油和了香酚具有很强的抑制酪氨酸硝化的能力,山苍子油、柠檬醛、香茅油、香茅醛仅在较高浓度时表现出较强的抑制蛋白质硝化的能力。
本文献摘自华中科技大学黄晓伟博士学位论文,如若侵权,请告知删除。
3.1 引言
酪氨酸酶是活性中心含有双核金属铜的氧化还原酶,是黑色素生物合成的关键酶[1]。组氨酸残基位于酪氨酸酶的内部,结合的铜离子为酪氨酸酶的活性位点[2.3]酪氨酸酶催化黑色素形成的前两步:L-酪氨酸羟化形成工-二羟苯丙氨酸(L-DOPA)和后续的联苯酚氧化为醌(L-多巴醌)[4-7]在催化反应过程中酪氨酸酶可以分为三种形态[8-10]:脱氧态(Edeoxy)、氧化态(Eoxy)和还原态(Emet)。还原型酶只具有二酚酶活性,氧化型酶同时具有酪氨酸羟化酶活性(即单酚酶活性)和多巴氧化酶活性(即二酚酶活性),而脱氧型酶能够结合氧分子。在酶促反应过程中,氧化型酶和还原型酶作用于底物的同时自身被还原成脱氧型,脱氧型酶能够结合一分子氧回到氧化状态,完成一个循环。酪酸酶二酚酶的活力就是酪氨酸酶在黑色素生成的第二步中表现出的活力。还原态的酶与L-DOPA 非共价结合形成醒,而还原态酶转化为脱态的酶的过程[11-12]。酪氨酸酶单酚酶活力通常被认为是酪氨酸酶在黑色素生成过程中第一步表现出来的活力,即 L-酪氨酸氧化成为L-DOPA 过程中表现出来的催化活性。但是实际上这·活力与酪氨酸酶氧化L-DOPA为醌的活力是同时表现的,所以研究学者通常将酪氨酸酶催化L-酪氨酸为L-DOPA,进一步催化为多巴醌的过程表现出来的酶活力称为单酚酶活力[11,13]。
在化妆品工业,越来越注重使用酪氨酸酶抑制剂来阻止黑色素的异常形成。酪氨酸酶抑制剂能够减少黑色素的合成,被用到化妆品中防止黑色素过度沉着,包括雀斑的形成。但是,只有极少数的美白剂被使用,典型的如曲酸和熊果苷,是目前商业上可以获得的。此外,有些美白剂的对细胞的高毒性、对氧和水的低稳定性导致其被限制使用。
在食品工业,胺类、氨基酸、肽类和蛋白质与醌(酶性褐变)之间的反应让食品在储藏或加工过程中容易变质。必须氨基酸的破坏、可消化性降低、蛋白水解酶和糖酵解酶的抑制是食品营养流失的主要原因。抗营养物质和毒性物质的产生会进一步降低食品的营养价值和安全性[14]。因此,建立多种阻止酶性褐变的方法至关重要。目前常用的防止褐变的方法有高压灭菌和短暂蒸煮的方法,这种传统的方法不可避免的造成营养物质的损失[15]。也有用微波技术来处理食品的方法,但是运用微波加热技术的最大限制因素是在使用微波的过程中会产生温度阶差[16]。酶的灭活需要高温处理,而低温是无法让酶完全失活。因此单纯使用微波加热技术无法让食物表面的酶完全失活。综合运用微波热水处理法能够在最终营养价值、食品重量、加工时间上有改进[17]。目前已经发现一些能够通过干扰酪氨酸酶介导的反应或者通过将醌类物质还原成酚来抑制食品的酶性褐变的化合物[18-20]。但是,因为这些化合物的气味、毒性和经济可行性使得真正能够在食品中运用的很少[21]。亚硫酸盐类物质曾经被广泛用于农业和海产品领域来防止酶性褐变,但由于其安全性问题被食品药品监督管理局禁用[22]。而食品工业中最多被运用的是维生素℃和柠酸[20,23]。但是他们防褐变的能力没有亚硫酸盐强,因为在还原酪氨酸酶形成的醌类物质的过程中维生素C会很快被消耗。4-已基间苯二酚在食品中的使用被认为是安全的,并且能够有效的防止新鲜的水果和水果干的褐变。但是,作为食品工业中使用的抑制剂,安全性是首要考虑的问题。因此,不断寻找天然来源的没有副作用的抑制剂意义重大。
综上所述,开发出一系列安全性高、能够广泛应用于食品及化妆品领域的酪氨酸酶抑制剂具有重要意义。本章以山苍子油、香茅油、丁香花蕾油为目标,研究其对酪氨酸酶的抑制活性,并对每种挥发油的主要成分做进一步酪氨酸酶抑制试验,确定挥发油中的活性成分。
3.2材料与方法
3.2.1试剂与仪器
3.2.1.1主要试剂
蘑菇酪氨酸酶、L-DOPA、曲酸: .上海华蓝试剂有限公司;柠檬醛、柠檬烯、芳樟醇、β-月桂烯、a-松油醇、a-蒎烯、β-蒎烯、丁香酚、乙酸丁香酯、香茅醛、香叶醇、香茅醇:阿拉丁T试剂公司; L -酪氨酸:国药集团化学试剂有限公二甲亚砜(DMSO)及其它化学试剂均为分析纯。试验用纯水均为18.2M9纯水。
3.2.1.2主要试验仪器设备
Milli-Q Synthesis超纯水系统:美国Millipore 公司;RT-6000酶标仪:深圳雷杜生命科学股份有限公司。
3.2.2酪氨酸酶抑制试验
首先将山苍子油、香茅油、丁香花蕾油和标准品(柠檬醛、柠檬烯、芳樟醇、β-月桂烯、a.-松油醇、a.-蒎烯、β-蒎烯、丁香酚、乙酸丁香酯、香茅醛、香叶醇、香茅醇)分别溶于DMSO,随后用磷酸盐缓冲溶液稀释至适当的浓度作为测试样品。
酪氨酸酶二酚酶活性的测定:按照之前文献报道的方法[24-28]并做适当的修改。L-DOPA作为酪氨酸酶活性测定的底物。500 μL pH 6.8的磷酸盐缓冲溶液,150 μL2.5mM L-DOPA,50 μL不同浓度的受试样品混合均匀,25°C孵育10分钟。然后加入25 μL蘑菇酪氨酸酶(1380U/mL)。25°C孵育10分钟后,立即测其在475nm处的吸光度值。曲酸作为阳性对照。试验重复3次,数据取平均值。
单酚酶活力的测定:先将300 μuL 3mmol/L的L-酪氨酸溶液与100 μuL磷酸盐缓冲液(pH6.8) 充分混匀,50 μL不同浓度的受试样品混合均匀,于30°C 水浴中温浴10 min后,加入100 μL蘑菇酪氨酸酶( 1380U/ mL)。25°C孵育10分钟后,立即在475 nm处测定吸光度值。曲酸作为阳性对照。试验重复3次,数据取平均值。
酪氨酸酶抑制率的计算:
Inhibition (%) = (Arest smple - Abank)/ Acontrol x 100
其中Acotol为混合液添加了酪氨酸酶而没有加入受试样品的吸光度值; Ablank为混合液加入受试样品但是没有加入酪氨酸酶的吸光度值; Atest sample是混合液加入了受试样品和酪氨酸酶后的吸光度值。
3.2.3柠檬醛抑制类型的测定
实验方法参照之前文献报道的方法并作适当修改[29, 30],不同浓度的L-DOPA(0.25 mM,0.333 mM,0.5 mM,1.0mM, 4.0 mM)作为底物, 825μL 磷酸盐缓冲溶液(pH6.8)加入75μL实验样品(0,0.1 mM, 0.5 mM),37.5μL 酪氨酸酶。用紫外分光光度计测475 nm处吸光值每分钟的增加量。酪氨酸酶活性的米氏常数Km和最大速度通过Lineweaver- _Burk plot来决定。
3.3实验结果
3.3.1丁香花蕾油与酪氨酸酶活性之间的关系
丁香花蕾油浓度与酪氨酸酶二酚酶活性之间的关系如图3.1A所示。我们选择具有强抑制酪氨酸酶活的曲酸为阳性对照,从图3.1A 我们可以看出,在曲酸浓度为0-20μg/m 时,随着曲酸浓度的增加,对酪氨酸酶二酚酶的活性有明显的抑制作用,抑制率达到 65.3%:当随着曲酸的浓度依次增高时,对酪氨酸酶二酚酶的抑制效率呈逐渐提升趋势,但增加的趋势较为缓慢,当曲酸浓度为100g/mL时,其对酪氨酸酶二酚酶的抑制率达到 76.1%。但丁香花蕾油对酪氨酸酶二酚酶的抑制率随其浓度的增加,抑制率呈负增长,表明丁香花蕾油对酪氨酸酶二酚酶没有明显的抑制作用。
为了进一步研究丁香花蕾油中的主要成分是否有抑制酪氨酸酶二酚酶酶活的能力,我们选取了丁香花蕾油中的三种主要成分,丁香酚、乙酸丁香酯和B-石竹烯,分别研究了他们的浓度和酪氨酸酶二酚酶抑制率之间的关系(图3.1B)。从图3.1B 中可以看出,丁香花蕾油主要成分丁香酚、乙酸丁香酯、B-石竹烯对酪氨酸酶二酚酶都没有明显的抑制能力。
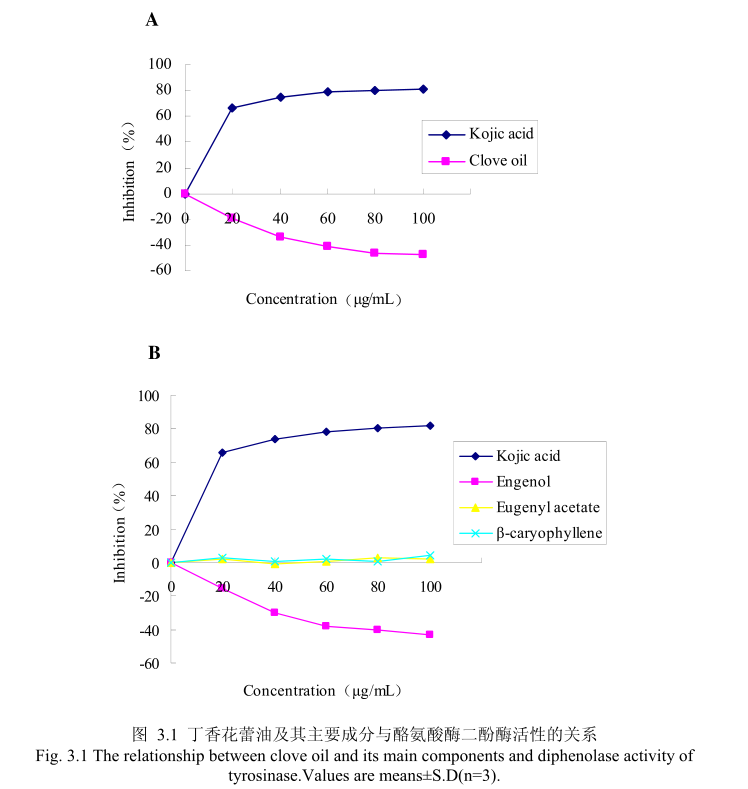
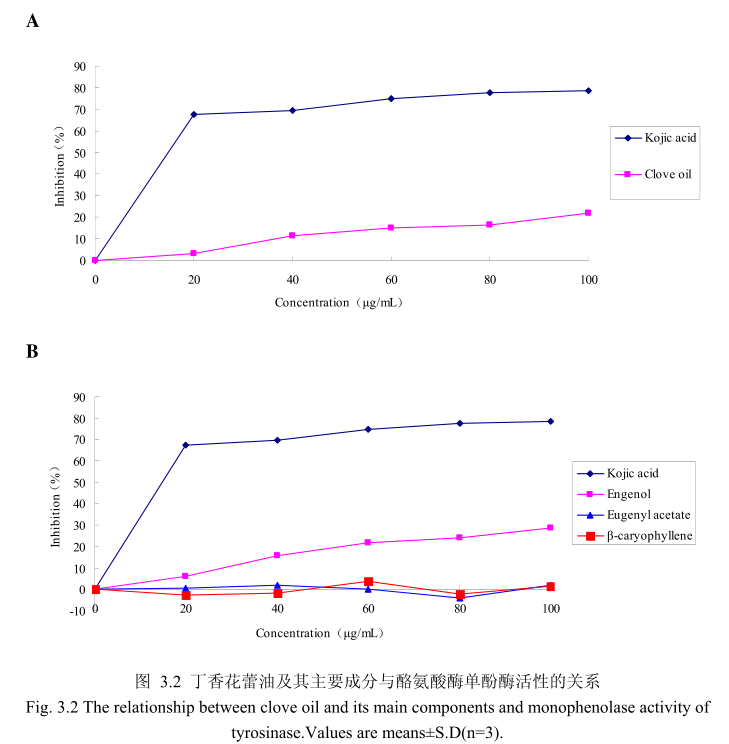
丁香花蕾油浓度与酪氨酸酶单酚酶活性之间的关系如图3.2A所示。我们选择具有强抑制酪氨酸酶活的曲酸为阳性对照,从图3.2A可以看出,在曲酸浓度为0-20μg/m 时,随着曲酸浓度的增加,对酪氨酸酶单酚酶活性有明显的抑制作用抑制率达到 70%;当随着曲酸的浓度依次增高时,对酪氨酸酶单酚酶的抑制率呈逐渐提升趋势,但增加的趋势较为缓慢,当曲酸浓度为100g/m时,其对酪氨酸酶单酚酶的抑制率达到 78.6%。丁香花蕾油对酪氨酸酶单酚酶的抑制率随其浓度的增加而增加,但是增加较为缓慢,当丁香花蕾油的浓度为100g/mL时,抑制率为 21.9%,表明丁香花蕾油对酪氨酸酶单酚酶活性有一定的抑制作用,但是曲酸对酪氨酸酶单酚酶活性的抑制作用强于丁香花蕾油。
为了进一步研究丁香花蕾油中的主要成分是否有抑制酪氨酸单酚酶的能力,我们选取了丁香花蕾油中的三种主要成分,丁香酚、乙酸丁香酯和B-石竹烯,分别研究了他们的浓度和酪氨酸酶单酚酶抑制率之间的关系(图3.2B)。从图3.2B中可以看出,丁香花蕾油主要成分丁香酚具有一定的抑制酪氨酸酶单酚酶活性的能力,当丁香酚的浓度为40μg/m 时,对酪氨酸酶单酚酶抑制率为16%,随着丁香酚的浓度逐渐增加,丁香酚对酪氨酸酶单酚酶的活性抑制作用逐渐增强,当丁香酚的浓度为 100g/m 时,丁香酚对酪氨酸酶单酚酶的活性的抑制率为28.6%。抑制作用比同样浓度下丁香花蕾油略强,但是比曲酸的的抑制作用弱。而丁香花蕾油的主要成分乙酸丁香酯、B-石竹烯对酪氨酸酶单酚酶都没有明显的抑制能力。
3.3.2 山苍子油与酪氨酸酶之间的关系
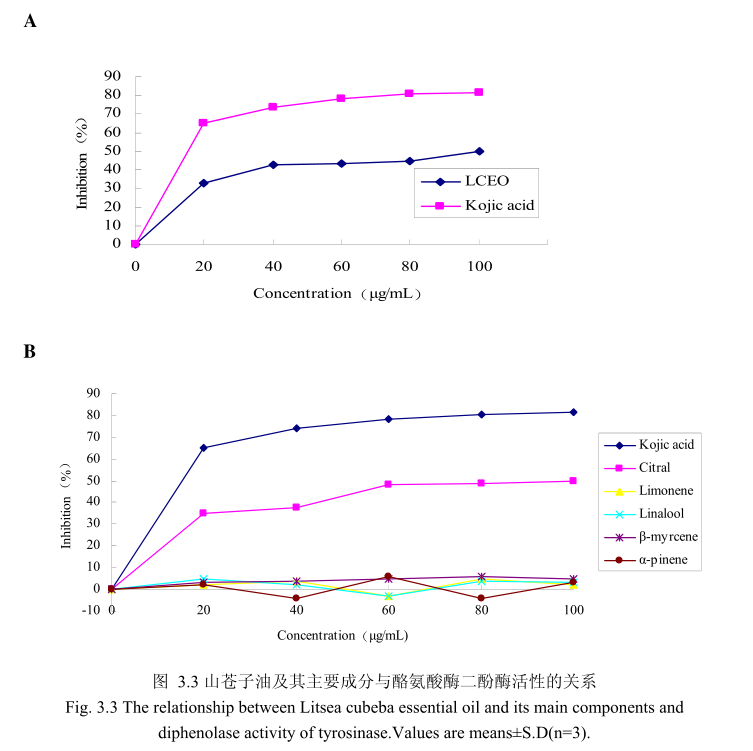
山苍子油浓度与酪氨酸酶二酚酶活性之间的关系如图3.3A 所示。我们选择具有强抑制酪氨酸酶活的曲酸为阳性对照,从图3.3A可以看出,在曲酸浓度为0-20Hg/m 时,随着曲酸浓度的增加,对酪氨酸酶二酚酶的活性有明显的抑制作用,抑制率达到 65.3%;当随着曲酸的浓度依次增高时,对酪氨酸酶二酚酶的抑制效率呈逐渐提升趋势,但增加的趋势较为缓慢,当曲酸浓度为100g/m时,其对酪氨酸酶二酚酶的抑制率达到 76.1%。山苍子油对酪氨酸酶二酚酶活性亦有明显的抑制作用,当浓度为0-20μg/m时,随着山苍子油浓度的增加,对酪氨酸酶二酚酶活性有明显的抑制作用,抑制率达到32%;当随着山苍子油的浓度依次增高时,对酪氨酸酶二酚酶活性的抑制率呈逐渐提升的趋势,但增加趋势较为缓慢,当山苍子油的浓度为100g/m 时,其对酪氨酸酶二酚酶的抑制率达到50%。因此,对照样品曲酸和山苍子油都对酪氨酸酶二酚酶的活性有较好的抑制作用,但是曲酸对酪氨酸酶二酚酶的抑制作用强于山苍子油。
为进一步研究山苍子油中的主要成分是否具有抑制酪氨酸酶二酚酶活性的能力,我们选取了山苍子油中的五种主要成分柠檬醛、柠檬烯、芳樟醇、月桂烯、松油醇,分别研究了他们的浓度和酪氨酸酶二酚酶抑制率之间的关系(图3.3B)。从图3.3B中可以看出,山苍子油主要成分柠醛对酪氨酸酶二酚酶活性具有较好的抑制能力,柠檬醛的浓度为 100μg/m 时,对酪氨酸酶二酚酶的抑制率达到 48%。而柠檬烯、芳樟醇、月桂烯、松油醇并没有表现出明显的对酪氨酸酶二酚酶的抑制作用。
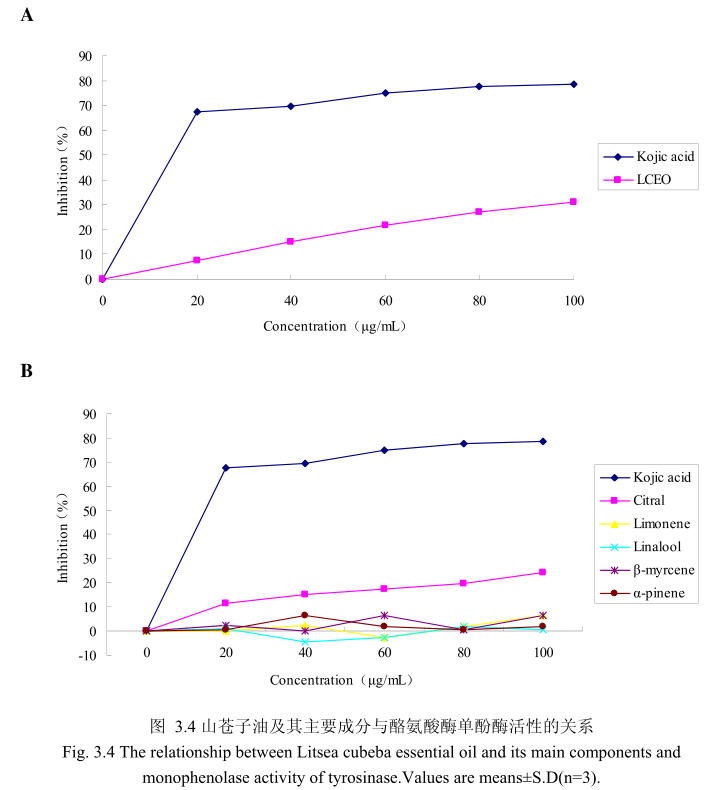
山苍子油浓度与酪氨酸酶单酚酶活性之间的关系如图3.4A所示。选择具有强抑制酪氨酸酶活性的曲酸为阳性对照。从图3.4A可以看出,在曲酸浓度为0-20μg/m 时,随着曲酸浓度的增加,对酪氨酸酶单酚酶的活性有明显的抑制作用,抑制率达到 67.4%;当随着曲酸的浓度依次增高时,对酪氨酸酶单酚酶的抑制率呈逐渐提升趋势,但增加的趋势较为缓慢,当曲酸浓度为100g/m时,其对酪氨酸酶单酚酶的抑制率达到 78.6%。山苍子油对酪氨酸酶单酚酶活性亦有一定的抑制作用,当浓度为0-20μg/mL 时,随着山苍子油浓度的增加,对酪氨酸酶单酚酶活性有一定的抑制作用,抑制率为7.6%;当随着山苍子油的浓度依次增高时,对酪氨酸酶单酚酶的抑制率呈逐渐提升的趋势,但增加趋势较为缓慢,当山苍子油的浓度为100g/m 时,其对酪氨酸酶单酚酶的抑制率达到31%。因此,对照样品曲酸对酪氨酸酶单酚酶的活性有较好的抑制作用,山苍子油对酪氨酸酶单酚酶具有一定的抑制作用。
为进一步研究山苍子油中的主要成分是否具有抑制酪氨酸酶单酚酶的能力,我们选取了山苍子油中的五种主要成分柠檬醛、柠檬烯、芳樟醇、月桂烯、松油醇,分别研究了他们的浓度和酪氨酸酶单酚酶抑制率之间的关系(图3.4B)。从图3.4B中可以看出,山苍子油主要成分柠檬醛对酪氨酸酶单酚酶活性具有一定的抑制能力,柠檬醛的浓度为100μg/m 时,对酪氨酸酶的抑制率达到 24.1%。而柠檬烯、芳樟醇、月桂烯、松油醇并没有表现出明显的对酪氨酸酶单酚酶的抑制作用。
3.3.3 柠檬醛抑制类型的测定
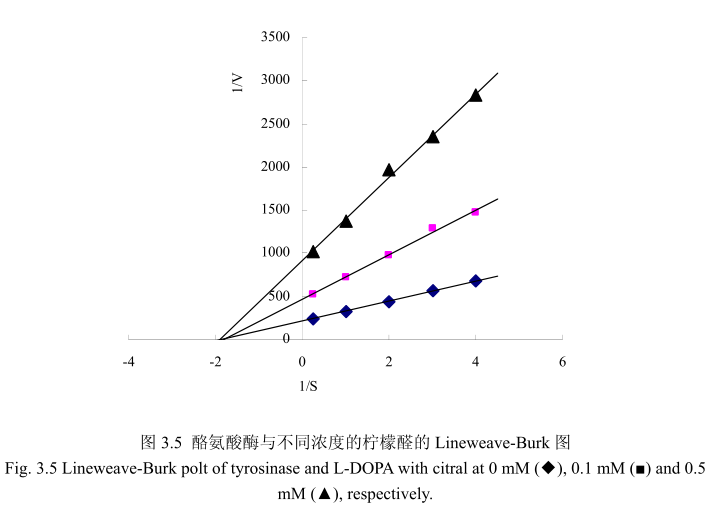
酶的抑制作用的研究主要是研究酶的催化机制、酶的结构和功能以及阐明酶的代谢途径的基本手段,同时也可以为新药物的设计、开发以及相关新产品研发提供一定的理论依据。因此,对酶的抑制作用及类型的研究具有重要的价值和理论意义如果抑制剂对酶的抑制作用是可逆的,即抑制剂与酶以非共价键的形式结合而引起酶的活力降低甚至是丧失,能够通过物理方法将抑制剂除去而让酶恢复活性,我们称之为可逆抑制;如果抑制剂对酶的抑制作用是不可逆的,即抑制剂与酶的必须基团以共价键的形式结合而引起酶失活,运用透析、超滤等物理方法不能将抑制剂去除而引起的酶活力丧失,我将其称之为不可逆抑制。可逆抑制作用可以根据抑制剂、底物和酶之间的关系而分为非竞争性抑制、反竞争性抑制和竞争性抑制。在非竞争性抑制过程中,抑制剂和底物能够同时和酶结合,两者之间没有竞争作用。酶与底物结合后还可以与抑制剂结合,同时酶与抑制剂结合后也可以与底物结合。但是这种由酶、抑制剂、底物结合而成的三元复合物不能够进一步分解为产物,因此会导致酶的活力降低。这种类型的抑制剂与酶的活性部位以外的基团相结合,其结构与底物没有相同的地方,这种类型的抑制作用不能通过增加底物浓度来解除抑制,所以称之为非竞争性抑制。在结构上,大多数的竞争性抑制剂与底物具有相似性,因此能够与酶的活性位点结合,与酶能够形成可逆的复合物,但是这种复合物不能够分解为产物,导致酶的反应速度下降。竞争性抑制剂的抑制程度取决于底物及抑制剂的相对浓度,这种抑制作用可以通过增加底物浓度而解除31。本节通过Lineweaver-Burk polts 分析抑制动力学,从而判断柠檬醛对酪氨酸酶抑制 L-DOPA氧化的机制。从图3.5可以看出,三条线分别代表不同浓度(0mM(◆),0.1mM(■),0.5 mM(▲))的柠檬醛,并且在 1/S 轴上相交于一点。结果表明柠檬醛是非竞争性抑制。结果与相关文献23报道一致。抑制剂柠檬醛与游离酶和酶底物复合物耦合醛类化合物如柠檬醛是蛋白质反应化合物,能够与生物学上重要的亲核基团基氨基或羟基反应。醛类能够与酪氨酸酶上的氨基形成席夫碱。不同醛类物质对酪氨酸酶的抑制活性的强弱取决于酪氨酸酶与醛类物质之间形成的席夫碱的稳定性。因此,可以认为酪氨酸酶抑制活性是由于柠檬醛与酪氨酸酶活性部位周围的亲核基团之间形成了席夫碱,形成了空间位阻阻碍酶与底物的结合,从而降低酶的活性。
3.3.4 香茅油与酪氨酸酶之间的关系
香茅油与酪氨酸酶二酚酶之间的关系如图3.6A所示。我们选择具有强抑制酪氨酸酶活性的曲酸为阳性对照。从图3.6A中可以看出,在曲酸浓度为0-20mg/mI时,随着曲酸浓度的增加,对酪氨酸酶二酚酶的活性有明显的抑制作用,抑制率达到 65.3%;当随着曲酸的浓度依次增高时,对酪氨酸酶二酚酶的抑制率呈逐渐提升趋势,但增加的趋势较为缓慢,当曲酸浓度为100g/m时,其对酪氨酸酶二酚酶的抑制率达到 76.1%。但是香茅油对酪氨酸酶二酚酶的抑制率随其浓度的增加,抑制率呈负增长,表明香茅油对酪氨酸酶二酚酶活性没有明显的抑制作用。
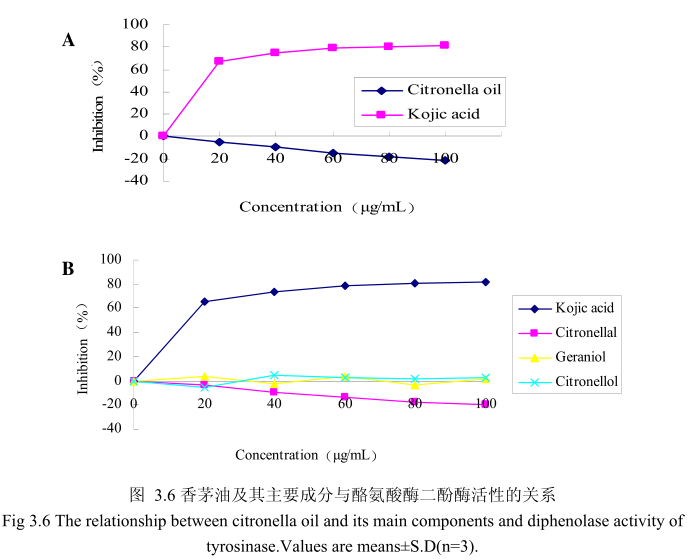
为了进一步研究香茅油中的主要成分是否有抑制酪氨酸酶二酚酶活性的能力,我们选取了香茅油中的三种主要成分,香茅醛、香叶醇和香茅醇,分别研究了他们的浓度和酪氨酸酶二酚酶抑制率之间的关系(图3.6B)。从图3.6B 中可以看出,香茅油主要成分香茅醛、香叶醇和香茅醇对酪氨酸酶二酚酶都没有明显的抑制能力。
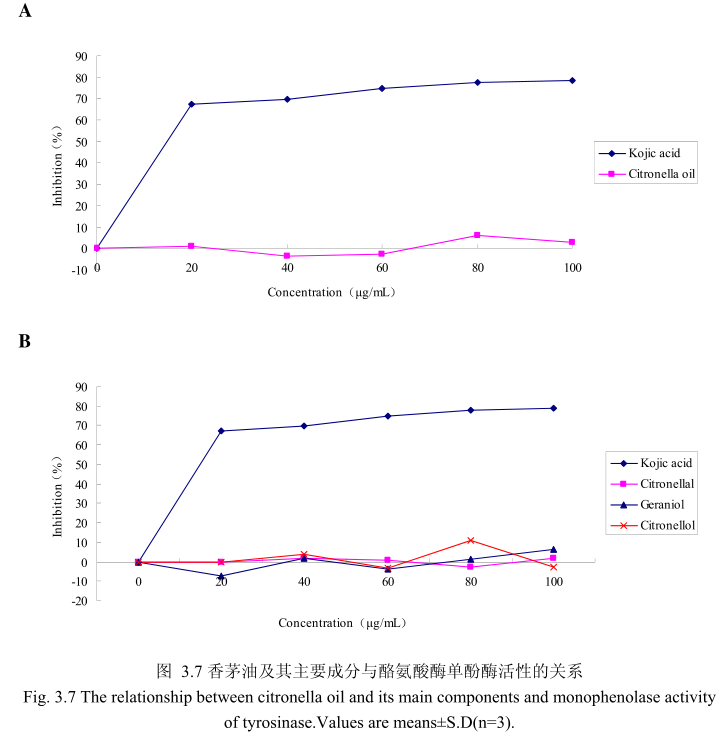
香茅油与酪氨酸酶单酚酶之间的关系如图3.7A所示。我们选择具有强抑制酪氨酸酶活的曲酸为阳性对照。从图3.7A中可以看出,在曲酸浓度为0-20g/mL时,随着曲酸浓度的增加,对酪氨酸酶单酚酶的活性有明显的抑制作用,抑制率达到 67.4%;当随着曲酸的浓度依次增高时,对酪氨酸酶单酚酶的抑制效率呈逐渐提升趋势,但增加的趋势较为缓慢,当曲酸浓度为100g/m时,其对酪氨酸酶单酚酶的抑制率达到 78.6%。但是香茅油对酪氨酸酶单酚酶活性的抑制率随其浓度的增加并没有明显的变化,表明香茅油对酪氨酸酶单酚酶活性没有明显的抑制作用。
为了进一步研究香茅油中的主要成分是否有抑制酪氨酸酶单酚酶活性的能力,我们选取了香茅油中的三种主要成分,香茅醛、香叶醇和香茅醇,分别研究了他们的浓度和酪氨酸酶抑制率之间的关系(图3.7B)。从图3.7B中可以看出,香茅油主要成分香茅醛、香叶醇和香茅醇对酪氨酸酶单酚酶都没有明显的抑制能力。
3.4 讨论
本章实验的主要目的是研究山苍子油、丁香花蕾油、香茅油对酪氨酸酶的抑制作用,并进一步探讨抑制机制。酪氨酸酶与人密切相关,是一种含有铜离子的酶通过两种不同的途径催化黑色素合成:通过酪氨酸酶羟化酪氨酸和氧化L-DOPA 为多巴配[32-34]。酪氨酸酶广泛存在于植物和动物中,参与黑色素的形成[35-37]。在食品工业,酪氨酸酶在控制食品和水果蔬菜的质量方面非常重要[36-38]。酪氨酸酶催化酚类物质氧化为对应的醌类,是水果和蔬菜酶性褐变的关键酶。此外,酶性褐变引起醌类物质与蛋白质上的氨基和巯基发生不可逆的反应,从而产生让人讨厌的颜色和气味。醌类物质与蛋白质的反应降低了蛋白质的可溶性和必须氨基酸的生物活性,包括赖氨酸和半胱氨酸。人类皮肤和头发的颜色取决于很多因素,最重要的就是黑色素的分布[39]。黑色素能够通过吸收紫外线和清除活性氧保护皮肤,但是黑色素的过度沉着如黑斑病、老年斑、雀斑常常影响美观。
挥发油通常具有明显的香气,常常用于香精的调配,广泛应用于食品和日化品中。同时,因为挥发油普遍具有抑制微生物生长的作用,能够作为天然防腐剂应用于食品及日化品中。而挥发油在养生保健方面,常常与植物油脂混合后直接用于皮肤按摩,不仅能够放松神经,更能起到养颜美容的功效。因此,如果挥发油能够作为酪氨酸酶抑制剂使用,将具有其它抑制剂所不具有的众多优势。
有相关文献报道丁香甲醇萃取物能够抑制B16黑素细胞中黑色素的形成[40]。丁香甲醇萃取物的主要活性物质为丁香酚和丁香乙酸酯,当浓度为100 μg/mL和200ug/mL时,对B16黑色素细胞中黑素的抑制率达到60%和40%。而丁香花蕾油同样含有丁香酚和丁香乙酸酯,当浓度为100μg/mL 和200 μg/mL时,对B16黑色素细胞中黑素的抑制率达到50%和80%。而本章实验中丁香花蕾油的主要成分为丁香酚和乙酸丁香酯,当其浓度均为100 μg/ mL时均没有表现出酪氨酸酶二酚酶抑制活性。丁香花蕾油对酪氨酸酶单酚酶的抑制率随其浓度的增加而增加,但是增加较为缓慢,当丁香花蕾油的浓度为100 ug / mL时,抑制率为219%,表明丁香花蕾油对酪氨酸酶单酚酶活性有一定的抑制作用。丁香花蕾油主要成分丁香酚具有一定的抑制酪氨酸酶单酚酶活性的能力,当丁香酚的浓度为40μg/ mL时,酪氨酸酶单酚酶抑制率为16%, 随着丁香酚的浓度逐渐增加,丁香酚对酪氨酸酶单酚酶的活性抑制作用逐渐增强,当丁香酚的浓度为100 μg/mL时,丁香酚对酪氨酸酶单酚酶活性的抑制率为28.6%。抑制作用比同样浓度下丁香花蕾油略强,但是比曲酸的抑制作用弱。而丁香花蕾油的其他主要成分如乙酸丁香酯、β-石竹烯对酪氨酸酶单酚酶都没有明显的抑制能力。体外实验与文献报道的细胞实验有一定的差异,可能是由于丁香花蕾油的酪氨酸酶抑制活性在体外实验环境和细胞实验环境中本身就存在差异。同时,由于体外实验环境是水溶性环境,丁 香花蕾油在水中的溶解度有限,如果当丁香花蕾油的浓度进--步增大,则表现出更强的酪氨酸酶抑制能力。
本实验中证明山苍子油及其主要成分柠檬醛对酪氨酸酶单酚酶和二酚酶活性均有抑制作用,与之前有文献报道柠檬油及其主要成分柠檬醛有酪氨酸酶抑制活性一致[29]。醛类是已知的与具有生物活性的亲核基团(如巯基、氨基、羟基)反应的重要基团,所以它的抑制活性是由于醛基可以与酪氨酸酶活性中心周围的氨基反应形成席夫碱,产生的空间位阻阻碍酶与底物的结合,从而酶活性降低。
鉴于试验中发现山苍子油的抑制效果要高于同等含量的柠檬醛,所以我们研究了山苍子油中主要成分对酪氨酸酶的抑制效果。从图3.3和3.4中可以看出,山苍子油对酪氨酸酶抑制的作用主要来自于柠檬醛,柠檬烯的作用很小,其他成分芳樟醇、松油醇、月桂烯在同等浓度下没有表现出对酪氨酸酶的抑制作用。可以推断,山苍子油所表现出对酪氨酸酶的抑制活性强于柠檬醛可能由于其他未知成分引起,也可能是由于在某种组分的协同作用下,使柠檬醛的抑制活性增强。
本章实验表明,香茅油及其主要成分香茅醛并没有表现出酪氨酸酶单酚酶和二酚酶的抑制活性。而香茅醛与柠檬醛有着相似的结构,并且醛类物质也可能与酪氨酸酶上的氨基形成席夫碱。但是香茅醛并没有表现出酪氨酸酶抑制活性,可能是由于香茅醛与酪氨酸酶.上的氨基形成的希夫碱稳定性比柠檬醛与酪氨酸酶上的氨基形成的席夫碱要弱。
3.5本章小结
本章主要探讨丁香花蕾油、山苍子油、香茅油对酪氨酸酶的抑制作用。通过实验发现,丁香花蕾油对酪氨酸酶二酚酶没有抑制作用,对酪氨酸酶单酚酶具有--定的抑制作用,当浓度为100 μg/mL时,抑制率为219%。丁香花蕾油的主要成分丁香酚、乙酸丁香酯、β-石竹烯对酪氨酸酶二酚酶没有抑制作用,但是J香酚对酪氨酸酶单酚酶具有- -定的抑制作用,当丁香酚的浓度为100 ug/mL时,丁香酚对酪氨酸酶单酚酶活性的抑制率为28.6%, 而乙酸丁香酯、β -石竹烯对酪氨酸酶单酚酶也没有表现出抑制作用。香茅油及其主要成分香茅醛、香叶醇、香茅醇对酪氨酸酶单酚酶和二酚酶均没有抑制作用。山苍子油对酪氨酸酶二酚酶有较好的抑制作用,当浓度为100 ug/ mL时对酪氨酸酶二酚酶活性的抑制率达到50%。山苍子油中的主要成分柠檬醛对酪氨酸酶二酚酶有抑制作用,当浓度为105μg/mL时对酪氨酸酶的活性抑制率达到50%,山苍子油中的其他主要成分柠檬烯、芳樟醇、月桂烯、松油醇并没有表现出明显的对酪氨酸酶__.酚酶的抑制作用。山苍子油对酪氨酸酶单酚酶活性亦有一定的抑制作用,当山苍子油的浓度为100 ug/mL时,其对酪氨酸酶单酚酶的抑制率达到31%。山苍子油中的主要成分柠檬醛对酪氨酸酶单酚酶活性具有一定的抑制能力,当柠檬醛的浓度为100 ug/ mL时,对酪氨酸酶单酚酶的抑制率达到24.1%。 而其他主要成分柠檬烯、芳樟醇、月桂烯、松油醇并没有表现出明显的对酪氨酸酶单酚酶的抑制作用。通过Lineweaver-Burk polts进一步分析柠檬醛的抑制类型,结果表明柠檬醛为非竞争性抑制。
3.6参考文献
[1] Kwon BS, Haq AK, Pomerantz SH, et al. Isolation and sequence of a cDNA clone for human tyrosinase that maps at the mouse c-albino locus. Proceedings of the National Academy of Sciences of the United States of America, 1987,84(21):7473- -7477
[2] Hearing VJ, Jimenez M. Mammalian tyrosinase - the critical regulatory control point in melanocyte pigmentation. International Journal of Biochemistry, 1987, 19(12):1141-1147
[3] Ito S, Wakamatsu K. Quantitative analysis of eumelanin and pheomelanin in humans, mice, and other animals: a comparative review. Pigment Cell Research, 2003, 16(5):523-531
[4] Prota G. The role of peroxidase in melanogenesis revisited. Pigment Cell Research, 1990,3:25-31
[5] Hearing V. Unraveling the melanocyte. American Journal of Human Genetics, 1993, 52(1):1-7
[6] Ito S, Fujita K, Takahashi H, et al. Characterization of melanogenesis in mouse and guinea pig hair by chemical analysis of melanins and of free and bound dopa and 5-S-cysteinyldopa. Journal of Investigative Dermatology, 1984, 83(1):12-14
[7] Strothkamp K, Jolley R, Mason H. Quaternary structure of mushroom tyrosinase. Biochemical and Biophysical Research Communications, 1976, 70(2):519-524
[8] 王芳.桑叶中酪氨酸酶抑制成分的研究.浙江工商大学博士学位论文.2008.
[9] Fenol LG, Rodriguez-Lopez JN, Garcia Sevilla F, et al. Analysis and interpretation of the action mechanism of mushroom tyrosinase on the monophenols and diphenols generating highly unstable O-quinones. Biochimica et Biophysica Acta(BBA)-Protein Structure and Molecular Enzymology, 2001,1548:1-22
[10] Decker H, Terwilliger N. Cops and robbers:putative evolution of copper oxygen-binding proteins. The Journal of Experimental Biology, 2000, 203:1777-1782
[11]林敏, 抑制剂对蘑菇酪氨酸酶与马铃薯多酚氧化酶抑制效应的比较研究.厦门大学硕士学位论文.2007.
[12] Naish-Byfield S, Riley PA. Oxidation of monohydric phenol substrates by tyrosinase. Biochemistry, 1992, 288:63-67
[13] Espin JC. Kinetic characterization of the substrate specificity and mechanism of mushroom tyrosinase. Biochemistry, 2000, 267:1270-1279
[14] Bos C, Gandichon C, Tome D. Nutritional and physiological criteria in the assessment of milk protein quality for humans. Journal of the American College of Nutrition, 2000,19(2): 191-205
[15] Konanayakam M, Sastry S. Kinetics of shrinkage of mushrooms during blanching. Journal of Food Science, 1988, 53(5): 1406-1411
[16] Maskan M. Drying, shrinkage and rehydration charateristics of kiwifruits during hot air and microwave drying. Journal of Food Engineering , 2001 ,48(2):177-182
[17] Devece C, Rodriguez-Lopez JN, Fenoll LG, et al. Enzyme inactivation analysis for industrial blanching applications: comparison of microwave, conventional, and combination heat treatments on mushroom polyphenoloxidase activity. Journal of Agricultural and Food Chemistry, 1999, 47(11):4506-4511
[18] Langdon T. Preventing of browning in fresh prepared potatoes without the use of sulfiting agents. Food Technology, 1987, 41(5):64-67
[19] Dudley ED, Hotchkiss JH. Cysteine as an inhibitor of polyphenol oxidase.Journal of Food Biochemistry, 1989, 13( 1):65-75
[20] Santerre CR, Cash J, Vannorman D. Ascorbic acid/citric acid combinations in the processing of frozen apple slices. Journal of Food Science, 1988, 53(6): 1713-1716
[21] Kim YJ, Uyama H. Tyrosinase inhibitors from natural and synthetic source:structure, inhibitor mechanism and perspective for the future. Cellular and Molecular Life Sciences, 2005,62:1707-1723
[22] Chalom s, Elrezzi E, Pena P, et al. Composition of sulfited potatoes: comparision with fresh and frozen potatoes. Plant Foods for Human Nutrition, 1995,47:133-138
[23] Hsu A, Shieh J, Bills D, et al. Inhibition of mushroom polyphenoloxidase by ascorbic acid derivatives. Journal of Food Science, 1988, 53(3):765-767
[24] Yu LL, Hu WC, Ding G, et al. Gusanlungionosides A-D, Potential Tyrosinase Inhibitors from Arcangelisia gusanlung. Jourmal of Natural Products, 2011,74(5): 1009-1014
[25] Kubo I, Kinst-Hori I. Tyrosinase inhibitors from cumin. Journal of Agricultural and Food Chemistry, 1998, 46( 12):5338-5341
[26] Kubo I, Kinst-Hori I, Chaudhuri SK, et al. Flavonols from Heterotheca inuloides: tyrosinase inhibitory activity and structural criteria. Bioorganic & Medicinal Chemistry, 2000, 8(7):1749- 1755
[27] Kubo I, Kinst-Hori I. Tyrosinase inhibitory activity of the olive oil flavor compounds. Journal of Agricultural and Food Chemistry, 1999,47(11):4574-4578
[28] Kubo I, Kinst: Hori I. Tyrosinase inhibitors from anise oil. Journal of Agricultural and Food Chemistry, 1998, 46(4):1268-1271
[29] Matsuura R, Ukeda H, Sawamura M. Tyrosinase inhibitory activity of citrus essential oils. Journal of Agricultural and Food Chemistry, 2006, 54(6):2309-2313
[30] Ha YM, Chung SW, Song S, et al. 4-(6-Hydroxy-2-naphthyl)-1, 3-bezendiol: apotent, new tyrosinase inhibitor. Biological and Pharmaceutical Bulletin, 2007,
30(9):1711-1715
[31] 王镜岩,朱圣庚,徐长法.生物化学.第三版.高等教育出版社,2002.368-369
[32] Canovas FG, Garcia-Carmona F, Sanchez JV, et al. The role of pH in the melanin biosynthesis pathway. Journal of Biological Chemistry, 1982, 257(15):8738-8744
[33] Neptuno Rodriguez-Lopez J, Tudela J, Varon R, et al. Kinetic study on the effect of pH on the melanin biosynthesis pathway. Biochimica et Biophysica Acta (BBA)-Protein Structure and Molecular Enzymology, 1991, 1076(3):379-386
[34] Cooksey CJ, Garratt PJ, Land EJ, et al. Evidence of the indirect formation of the catecholic intermediate substrate responsible for the autoactivation kinetics of tyrosinase. Jourmal of Biological Chemistry, 1997,272(42):26226-26235
[35] Pawelek JM, Korner AM. The Biosynthesis of Mammalian Melanin: The regulation of pigment formation, the key to disorders such as albinism and piebaldism, may also offer some clues for the treatment of melanoma. American Scientist, 1982, 70(2):136-145
[36] Mayer AM. Polyphenol oxidases in plants -recent progress. Phytochemistry, 1986, 26(1):11-20
[37] Kocher A, Choct M, Hughes RJ, et al. Effect of food enzymes on utilisation of lupin carbohydrates by broilers. British Poultry Science, 2000,41(1):75-82
[38] Friedman M. Food browning and its prevention: an overview. Journal of Agricultural and Food Chemistry, 1996, 44(3):631-653
[39] Passeron T, Mantoux F, Ortonne JP. Genetic disorders of pigmentation. Clinics in Dermatology, 2005,23(1):56-67
[40] Arung ET, Matsubara E, Kusuma IW, et al. Inhibitory components from the buds of clove (Syzygium aromaticum) on melanin formation in B 16 melanoma cells. Fitoterapia, 2011, 82(2):198-202
未完待续……
2.1 引言
挥发油是芳香植物中次生代谢形成的具有挥发性的天然混合物,具有强烈的香气。挥发油最初在中世纪由阿拉伯人通过水蒸气蒸馏获得。由于其具有杀菌、抗病毒、抑制微生物繁殖的特性,被用于食品保存、抗微生物、止痛、镇静、抗炎、解痉挛和局部麻醉。直至今天,这些性质并没有很大变化,只是在作用机制方面的研究更加深入,尤其是在抗微生物领域的研究。
在自然界,挥发油在保护植物方面有着重要的作用,如抗微生物、抗病毒、抗真菌、杀虫、降低食草动物的食欲。
挥发油为传统药典中的一个重要部分,外观为澄清液体,具有挥发性,通常密度小于水。所有的植物器官都能提取挥发油,如花蕾、花、叶、茎、小分支、种子果实、根、树干或树皮。提取挥发油的方法有多种,如超临界流体取、微波辅助萃取、水蒸气蒸馏、有机溶剂萃取。由于挥发油具有抗微生物和杀灭真菌的活性能够取代化学杀菌剂而在制药和食品领域得到越来越广泛的应用。在这种情况下,水蒸气蒸馏法和压榨法常被使用,如柑橘挥发油的提取。在香精领域,常常运用超临界流体萃取和水蒸气蒸馏法提取挥发油。提取的挥发油会因为植物的生长气候、土壤环境、植物器官、生长年龄和周期的不同而在挥发油含量、质量和化学成分上存在差异[1-2]。因此,为了获得成分基本一致的挥发油,通常选择植物的相同部位进行提取,且这些植物的土壤环境一样、生长气候一样、采摘季节一样。许多商品化的挥发油都要用气相色谱和质谱联用来分析成分。通过分析报告来保证挥发油的质量[3]
本章采用提取挥发油最常用的水蒸气蒸馏法,提取丁香花蕾油、香茅油、山苍子油,并用GC、GC-MS分析每种挥发油的成分,确定各种油的主要成分的相对含量。
2.2 材料和方法
2.2.1 主要原料及试剂
丁香花蕾:2009年采摘于中国广东,室温条件下自然风干;香茅:2009年采摘于中国广东,室温条件下自然风干;山苍子果实:2009年采摘于中国广东,室温条件下自然风干;无水硫酸钠(国药集团化学试剂有限公司);正构烷烃C8~C40(北京百灵威科技有限公司)。
2.2.2 主要仪器
气质联用仪(ThermoTrace GC/Ultra-DSQI );油水分离器(武汉大学科教仪器厂);气相色谱仪(Thermo Trace GC ULTRA);密度折光联用仪(Antonpaar股份有限公司);电热套(巩义市予华仪器有限公司)。
2.2.3 挥发油提取的单因素条件实验
山苍子挥发油提取单因素条件实验:选择优质的山苍子,提取的粒径选择未粉碎、粉碎至20目、粉碎至40目、粉碎至60目、粉碎至80目五个水平;浸泡时间分别采用0小时、2 小时、4 小时、6 小时、8 小时、10 小时和 12小时七个水平;料液比 1:4、1:6、1:8、1:10、1:12、1:14六个水平;提取时间为2小时、4小时、6 小时、8小时、10小时五个水平。分别进行单因素实验
丁香花蕾挥发油提取单因素条件实验:选择优质的丁香花蕾,提取的粒径选择未粉碎、粉碎至 20目、粉碎至40目、粉碎至60目、粉碎至80目五个水平;浸泡时间采用0小时、2 小时、4 小时、6 小时、8 小时、10 小时、12 小时七个水平;料液比 1:4、1:6、1:8、1:10、1:12、1:14 六个水平;提取时间为 2小时、4 小时、6 小时、8 小时、10 小时五个水平。分别进行单因素实验。
香茅叶挥发油提取单因素条件实验:选择优质的香茅叶,提取的粒径选择整叶、剪碎、粉碎至20目、粉碎至40目、粉碎至60目5个水平;浸泡时间采用0小时、2 小时、4 小时、6 小时、8 小时五个水平;料液比 1:4、1:6、1:8、1:10、1:12、1:14 六个水平;提取时间为 2 小时、4 小时、6 小时、8 小时、10小时五个水平。分别进行单因素实验。
挥发油得率(%)=挥发油质量(g)/提取原料质量(g)x100%
2.2.4 最优条件下水蒸气蒸馏法提取挥发油
三种植物挥发油的提取如文献报道[4.5],只做了少量的修改,具体如下所述:山苍子油:取山苍子果300克粉碎,过筛,放入挥发油提取装置,加入10倍质量的1%的氯化钠溶液,浸泡8小时后,水中蒸馏6小时,油水分离器中收集山苍子油。向山苍子油中加入适量的无水硫酸钠,静置2小时,过滤,得到的挥发油称重,然后在 4℃ 冷藏柜中密封避光保存待用。
丁香花蕾油:取丁香花蕾300克粉碎,过,放入挥发油提取装置,加入12倍质量的1%的氯化钠溶液,浸泡6小时后,水中蒸馏6小时,油水分离器中收集丁香花蕾油。向丁香花蕾油中加入适量的无水硫酸钠,静置2小时,过滤,得到的挥发油称重,然后在4℃冷藏柜中密封避光保存待用。
香茅油:取香茅叶300克粉碎,过,放入挥发油提取装置,加入8倍质量的1%的氯化钠溶液,浸泡4小时后,水中蒸馏4小时,油水分离器中收集香茅油。向香茅油中加入适量的无水硫酸钠,静置2小时,过滤,得到的挥发油称重,然后在4℃冷藏柜中密封避光保存待用。
2.2.5 挥发油成分分析
GC-MS 分析条件:
毛细管柱:HP5-MS(30mx0.25mmi.d.x0.25 um d.f);进样口温度:250℃;升温程序:起始柱温50℃ ,保持2min,5℃min 升温至250℃,保持15min;载气:He,流速 1.0mL/min;样品用无水乙醇稀释100倍;进样量:1μL,分流比 50:1;质谱接口温度:250℃:电离方式:EI:电离能量:70eV:离子源温度:250℃:质量扫描范围:41-350amu。采用Wiley和Nist谱检索、人工图谱解析以及查对质谱相关文献进行定性。正构烷烃C~C的混合标样用相同的分析条件分析来计算保留指数。
GC-FID 分析条件:
GC-FID分析条件与GC-MS相似,但是载气为氮气。挥发油各组分的相对含量在没有使用矫正因子的条件下通过GC-FID峰面积计算而来。
2.3 实验结果
2.3.1 水蒸气蒸馏法提取挥发油工艺优化
2.3.1.1 水蒸气蒸馏法提取山苍子挥发油工艺优化
2.3.1.1.1 不同提取粒径对山苍子挥发油得率的影响
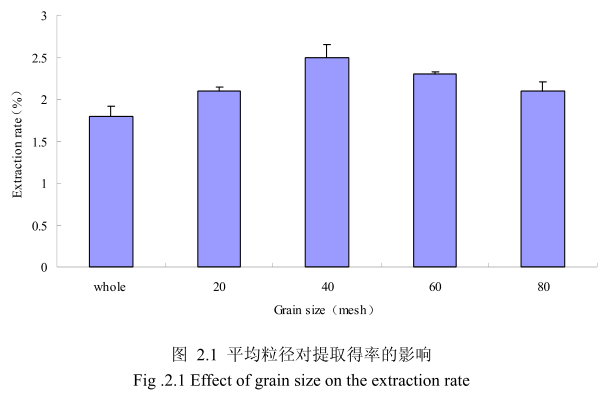
选择提取浸泡时间6小时、料液比1:8(质量比)、提取时间6小时、提取的粉碎粒径为未粉碎、粉碎至20目、粉碎至40目、粉碎至60目、粉碎至80目进行提取实验,实验结果如图2.1所示。从图中可以看出,当山苍子的提取粒径从未粉碎、粉碎至20目到粉碎至40目的过程中,山苍子油的得率随着粉碎粒径的变细而增大,当粉碎粒径为40目时,挥发油的得率最高。该过程中由于山苍子粉碎的越细,比表面积就越大,越有利于挥发油的浸出,所以挥发油的得率随着粉碎粒径的变小而增大。但是,当山苍子的粉碎粒径变为60目和80目时,山苍子油的得率反而下降。这可能是由于粉碎粒径的变小让颗粒比表面积增大的同时,颗粒自身的表面吸附作用增加,从而抑制了挥发油的扩散。同时,挥发油具有较强的挥发性,当粉碎粒径过细时,粉碎过程中会有少量的挥发油挥发损失掉,从而降低了挥发油的最终得率。因此,从本实验可以看出,山苍子的提取平均粒径控制在40目时挥发油的得率最高。
2.3.1.1.2 不同浸泡时间对山苍子挥发油得率的影响
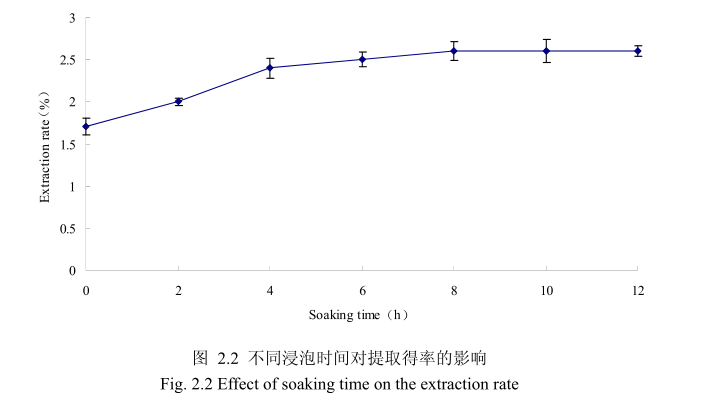
选择提取粒径为40目、料液比1:8(质量比)、提取时间6小时、提取的浸泡时间为0小时、2 小时、4 小时、6 小时、8 小时、10 小时、12 小时进行提取实验,从上图2.2可以看出,浸泡时间在0到8小时时间范围内,山苍子挥发油提取率随着浸泡时间的延长而增大。当浸泡时间为8小时,挥发油的得率达到较大值,随着浸泡时间的进一步延长,挥发油的得率几乎没有变化。造成这种现象的原因可能是由于浸泡过程可以使植物的细胞间隙增大,细胞组织充分膨胀,从而加速细胞内外的液体交换与平衡。由于山苍子质地较为坚硬,吸水性较差,因此需要浸泡较长时间才能让细胞组织充分膨胀。但是当浸泡达到一定时间后,细胞内外液体交换已经达到平衡,所以浸泡时间的进一步延长并不能改变精油的提取率。因此,选择浸泡时间为8小时时提取效率最高。
2.3.1.1.3 不同料液比对山苍子挥发油得率的影响
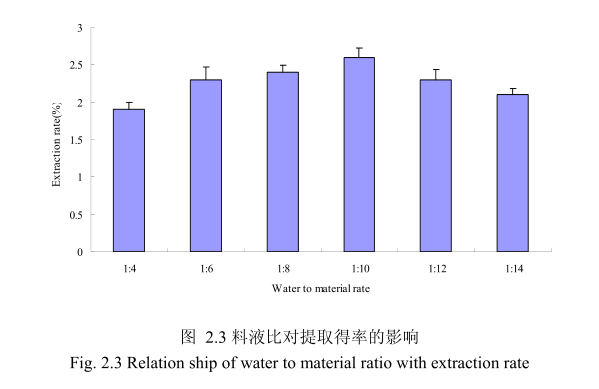
选择提取粒径40目,浸泡时间8小时,提取时间6小时,料液比为 1:4、1:6、1:8、1:10、1:12、1:14进行单因素实验。实验结果如图 2.3 所示,从图中可以发现,料液比从 1:4变化到1:10的过程中山苍子油的得率逐渐增加,料液比为1:10时山苍子油的得率最大。得率增加的原因可能是由于水的增多使山苍子中的山苍子油与水接触面处的浓度差变大,有利于山苍子油从组织中扩散出来。但当料液比从1:10 变化到 1:14 的过程中山苍子油的得率反而逐渐减少。可能由于水的增加,造成挥发油在水中的损耗增加,但是水量增加到一定程度对丁香花蕾油的扩散作用的影响已经不大,所以挥发油的得率降低。因此,当料液比为1:10时山苍子油的得率最高。
2.3.1.1.4 不同提取时间对山苍子挥发油得率的影响
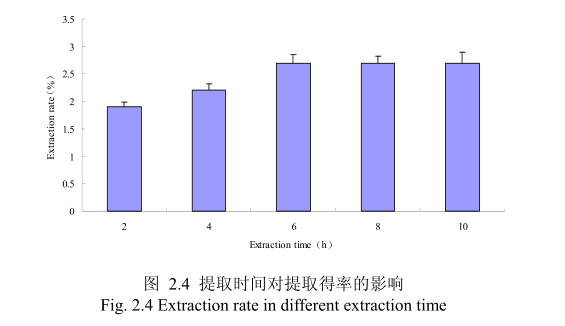
选择提取粒径为40目、料液比1:10(质量比)、浸泡时间8小时、提取时间为2小时、4 小时、6 小时、8小时、10小时进行提取实验,从图 2.4 中可以看出随着提取时间的不断延长,山苍子油的得率显著提高,当提取时间达到6小时时,山苍子油的得率达到较大值,随着提取时间的进一步延长,山苍子油的得率并没有显著的变化。所以可以推测,当山苍子提取到6小时时,体系的渗透压达到平衡,提取率趋于恒定,随着加热时间的延长,挥发油的提取效率并不能提高。
2.3.1.2 水蒸气蒸馏法提取丁香花蕾挥发油工艺优化
2.3.1.2.1 不同提取粒径对丁香花蕾挥发油得率的影响
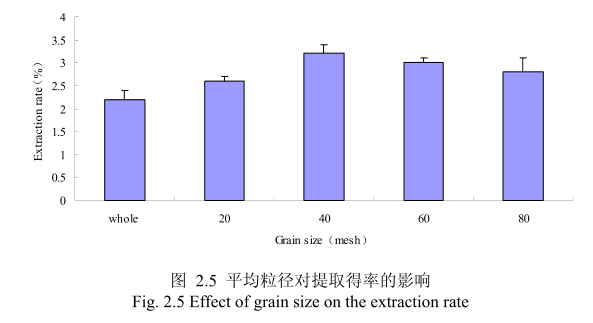
选择提取浸泡时间6小时、料液比1:8(质量比)、提取时间6小时、丁香花蕾的粉碎粒径分别为未粉碎、20目、40目、60目、80 目时进行提取实验,实验结果如图2.5所示。从图中可以看出,当丁香花蕾的提取粒径从未粉碎、粉碎至20目到粉碎至40目的过程中,丁香花蕾油的得率随着粒径的变细而增大,当粉碎粒径为 40目时,挥发油的得率最高。该过程中由于丁香花蕾粉碎的越细,比表面积就越大,越有利于挥发油的浸出,所以挥发油的得率随着粉碎粒径的变小而增大。但是,当丁香花蕾的粉碎粒径变为60目和80目时,丁香花蕾挥发油的得率反而下降。这可能是由于粉碎粒径的变小让颗粒比表面积增大的同时,颗粒自身的表面吸附作用增加,从而抑制了挥发油的扩散。同时,挥发油具有较强的挥发性当粉碎粒径过细时,粉碎过程中会有少量的挥发油挥发损失掉,从而降低了挥发油的最终得率。因此,从本实验可以看出,丁香花蕾的提取平均粒径控制在40目时挥发油的得率最高。
2.3.1.2.2 不同浸泡时间对丁香花蕾挥发油得率的影响
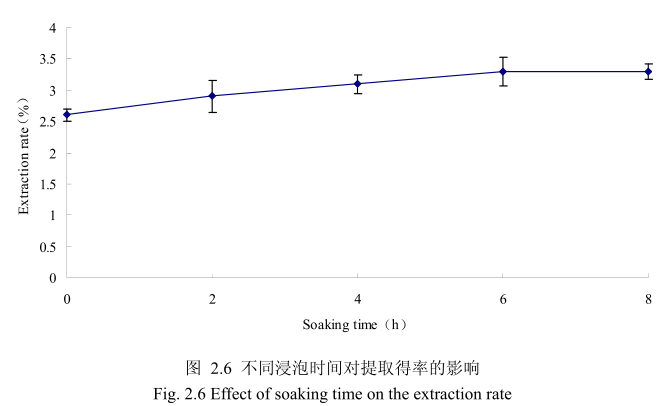
选择丁香花蕾粒径为40目、料液比1:8(质量比)、提取时间6小时。提取的浸泡时间分别为0小时、2小时、4 小时、6小时、8 小时进行提取实验,从图 2.6可以看出,浸泡时间在0到6小时时间范围内,丁香花蕾挥发油提取率随着浸泡时间的延长而增大。当浸泡时间为6小时时,挥发油的得率达到较大值,随着浸泡时间的进一步延长,挥发油的得率几乎没有变化。造成这种现象的原因可能是由于浸泡过程可以使植物的细胞间隙增大,细胞组织充分膨胀,从而加速细胞内外的液体交换与平衡。但是当浸泡达到一定时间后,细胞内外液体交换已经达到平衡,所以浸泡时间的进一步延长并不能改变挥发油的提取率。因此,选择浸泡时间为6小时时提取效率最高。
2.3.1.2.3 不同料液比对丁香花蕾挥发油得率的影响
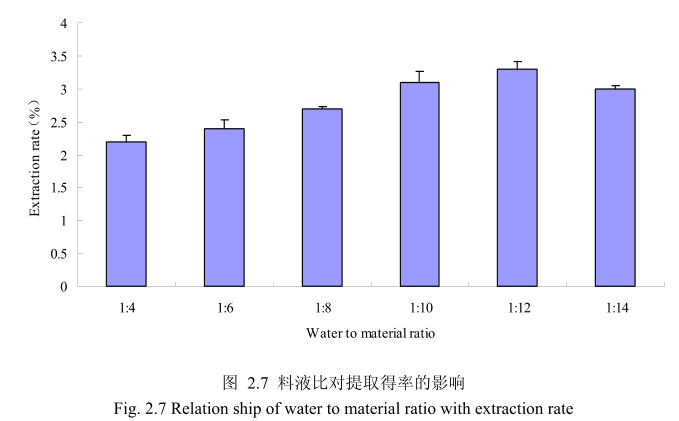
选择丁香花蕾粒径40目,浸泡时间6小时,提取时间6小时,料液比为1:4、1:6、1:8、1:10、1:12、1:14进行单因素实验。实验结果如图 2.7 所示,从图中可以发现,料液比从1:4变化到1:12的过程中丁香花蕾油的得率逐渐增加,料液比为1:12时丁香花蕾油的得率最大。得率增加的原因可能是由于水的增多使丁香花蕾中的丁香花蕾油与水接触面处的浓度差变大,有利于丁香花蕾油从组织中扩散出来。但当料液比从1:12变化到1:14的过程中,丁香花蕾油的得率反而逐渐减少。可能由于水的增加,造成挥发油在水中的损耗增加,但是水量增加到一定程度对了香花蕾油的扩散作用的影响已经不大,所以挥发油的得率降低。因此,当料液比为1:12时丁香花蕾油的得率最高。
2.3.1.2.4 不同提取时间对丁香花蕾挥发油得率的影响
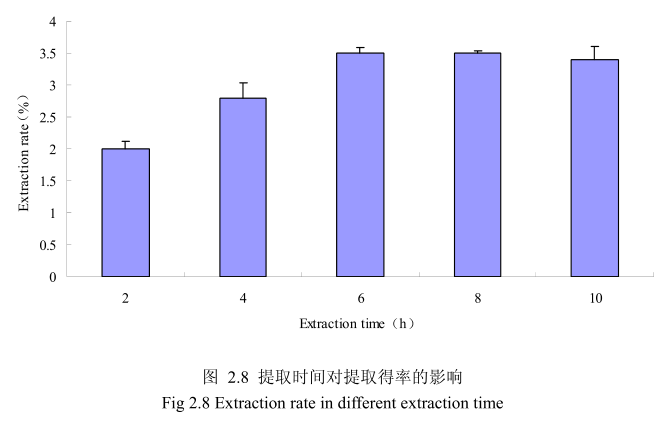
选择丁香花蕾粒径为40目、料液比1:12(质量比)、浸泡时间6小时、提取时间为2小时、4 小时、6小时、8小时、10 小时进行提取实验,从图2.8中可以看出随着提取时间的不断延长,丁香花蕾挥发油的得率显著提高,当提取时间达到6小时时,丁香花蕾挥发油的得率达到较大值,随着提取时间的进一步延长,丁香花蕾挥发油的得率并没有显著的变化。所以可以推测,当丁香花蕾提取到6时时体系的渗透压达到平衡,提取率趋于恒定,随着加热时间的延长,挥发油的提取效率并不能提高。
2.3.1.3 水蒸气蒸馏法提取香茅叶挥发油工艺优化
2.3.1.3.1 不同提取粒径对香茅叶挥发油得率的影响
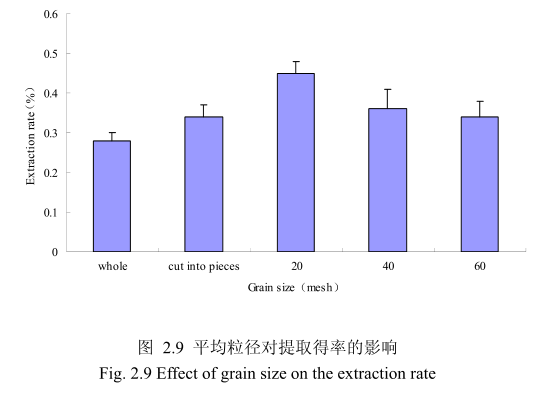
不同提取粒径对香茅叶挥发油的提取得率有一定的影响。选择提取浸泡时间6小时、料液比 1:10(质量比)、提取时间6小时、提取的粉碎粒径为整叶、剪碎、粉碎至20目、粉碎至40目、粉碎至60目进行提取实验,实验结果如图2.9所示。从图中可以看出,当香茅叶的提取粒径从整叶、剪碎到粉碎至 20目的过程中,香茅油的得率随着粉碎粒径的变细而增大,当粉碎粒径为20目时,挥发油的得率最高。该过程中由于香茅叶粉碎的越细,比表面积就越大,越有利于挥发油的浸出,所以挥发油的得率随着粉碎粒径的变小而增大。但是,当香茅叶的粉碎粒径变为40目和60目时,香茅挥发油的得率反而下降。这可能是由于粉碎粒径的变小让颗粒比表面积增大的同时,颗粒自身的表面吸附作用增加,从而抑制了挥发油的扩散。同时,挥发油具有较强的挥发性,当粉碎粒径过细时,粉碎过程中会有少量的挥发油挥发损失掉,从而降低了挥发油的最终得率。因此,从本实验可以看出,香茅叶提取平均粒径控制在20目时挥发油的得率最高。
2.3.1.3.2 不同浸泡时间对香茅叶挥发油得率的影响
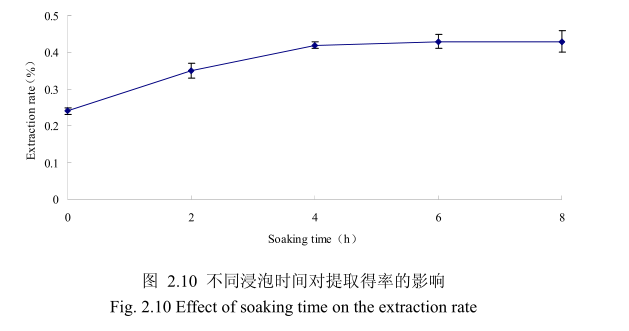
选择香茅叶粒径为20目、料液比1:10(质量比)、提取时间6小时、提取的浸泡时间分别为0小时、2小时、4 小时、6 小时、8 小时进行提取实验。从图2.10可以看出,在0到4时时间范围内,香茅叶挥发油提取率随着浸泡时间的延长而增大,在4小时时,挥发油的得率达到较大值,随着浸泡时间的进步延长,挥发油的得率几乎没有变化。造成这种现象的原因可能是由于浸泡过程可以使植物的细胞间隙增大,细胞组织充分膨胀,从而加速细胞内外的液体交换与平衡。但是当浸泡达到一定时间后,细胞内外液体交换已经达到平衡,所以浸泡时间的进一步延长并不能明显改变挥发油的提取率。因此,选择浸泡时间为4小时,此时提取效率最高。
2.3.1.3.3 不同料液比对香茅叶挥发油得率的影响
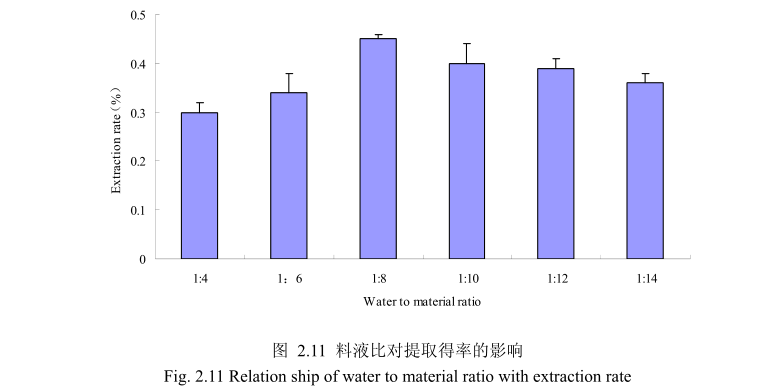
选择香茅叶粒径20目,浸泡时间4小时,提取时间6小时,料液比为1:4、1:6、1:8、1:10、1:12、1:14进行单因素实验。实验结果如图,从图中可以发现,料液比从1:4变化到1:8的过程中香茅叶油的得率逐渐增加,料液比为1:8时香茅叶油的得率最大。得率增加的原因可能是由于水的增多使香茅叶中的香茅油与水接触面处的浓度差变大,有利于香茅油的从叶片中扩散出来。但当料液比从1:8变化到1:14的过程中香茅油的得率反而逐渐减少。可能由于水的增加,造成挥发油在水中的损耗增加,但是水量增加到一定程度对香茅油的扩散作用的影响已经不大,所以挥发油的得率降低。因此,当料液比为1:8时香茅油的得率最高。
2.3.1.3.4 不同提取时间对香茅叶挥发油得率的影响
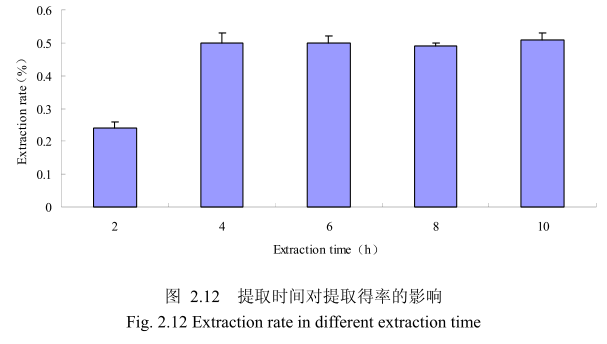
选择香茅叶粒径为20目、料液比1:8(质量比)、浸泡时间4小时、提取时间分别为 2小时、4 小时、6 小时、8 小时、10 小时进行提取实验,从图 2.12中可以看出随着提取时间的不断延长,香茅叶油的得率不断提高,当提取时间达到4小时时,香茅叶油的得率达到较大值,随着提取时间的进一步延长,香茅油的得率并没有显著的变化。所以可以推测,当香茅叶提取到4小时时,体系的渗透压达到平衡,提取率趋于恒定,随着加热时间的延长,挥发油的提取效率并不能提高。因此,当提取时间为4小时时,香茅油得率最高。
2.3.2 三种植物挥发油的物理指标
(1)丁香花蕾油:得率3.5%,澄清淡黄色液体,辛香及丁香酚的特征香气;相对密度 1.045(20/20℃);折光指数 1.5290(20℃)。
(2)山苍子油:得率2.7%,澄清淡黄色液体;感官特征:具有清鲜、甜的果香,类似柠檬油的香气;相对密度0.889(20/20℃);折光指数1.482(20℃)。
(3)香茅油:得率0.5%,澄清淡黄色液体;感官特征:具有轻快、新鲜的清甜果香,有成熟柠檬果皮的香气;相对密度0.889(20/20℃);折光指数 1.486(20℃)。
2.3.3 三种植物挥发油化学成分分析
2.3.3.1 山苍子油的化学成分
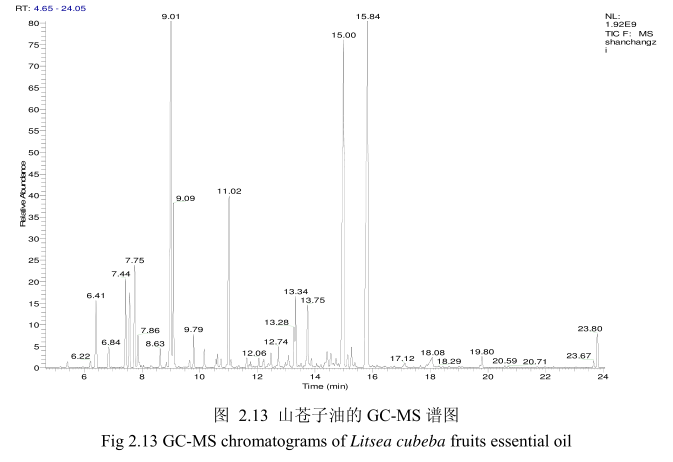
将最优提取条件下提取的山苍子油进行GC-MS分析。图2.13是山苍子油的GC-MS 图谱。从图2.13可以看出,山苍子油的化学成分较多,其中大多成分集中在保留时间5.0-25分钟之间。通过和标准品或质谱数据库数据比对,鉴定出19种化学成分,占总油含量的92.4%。表2.1为山苍子油成分和含量统计表。从表2.1可看出,广东产的山苍子果挥发油中相对含量较高的成分有E-柠醛(30.7%);Z-柠檬醛(26.7%);柠檬烯(8.1%);芳樟醇(5.2%);B-月桂烯(4.7%);a-松油醇(3.9%);B-蒎烯(2.7%);a-蒎烯(1.9%);香叶醇(1.3%);1.8-桉叶素(1.2%)等。

2.3.3.2 丁香花蕾油的化学成分
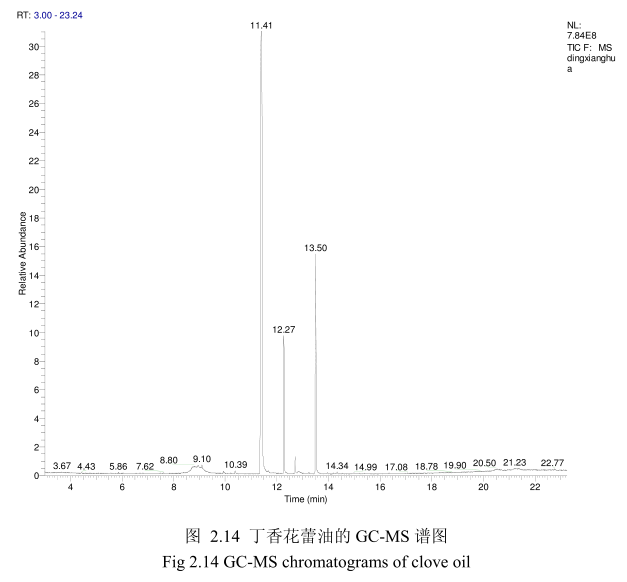
将最优提取条件下提取的丁香花蕾油通过GC-MS分析。图2.14是丁香花蕾油的GC-MS 图谱。从图2.14可以看出,丁香花蕾油的化学成分大多集中在保留时间 3.5-25分钟之间。通过和标准品或质谱数据库数据比对,鉴定出10种化学成分,占总油含量的94.8%。表2.2为鉴定出的丁香花蕾油成分和含量。从表2.2可看出,广东产的丁香花蕾油中相对含量较高的成分有丁香酚(76.8%)、乙酸丁香酯 (9.5%)、B-石竹烯(6.0%)、E-3-十二烯(1.9%)、2-1-十二烯(1.3%)等。
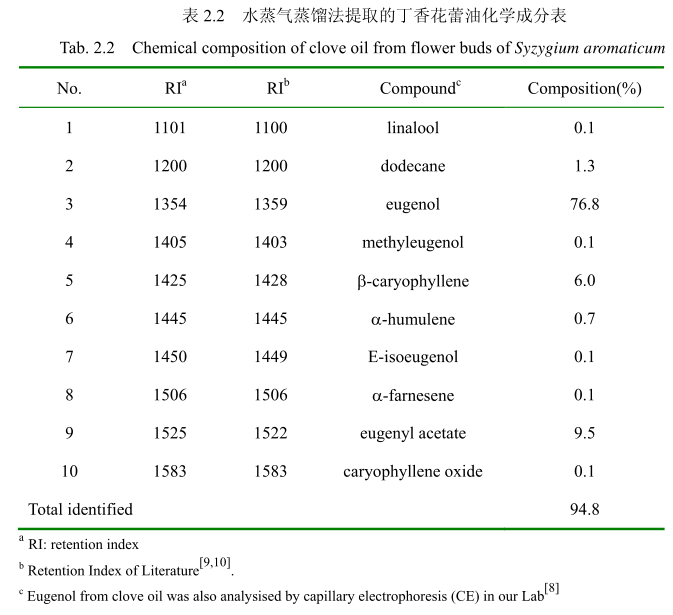
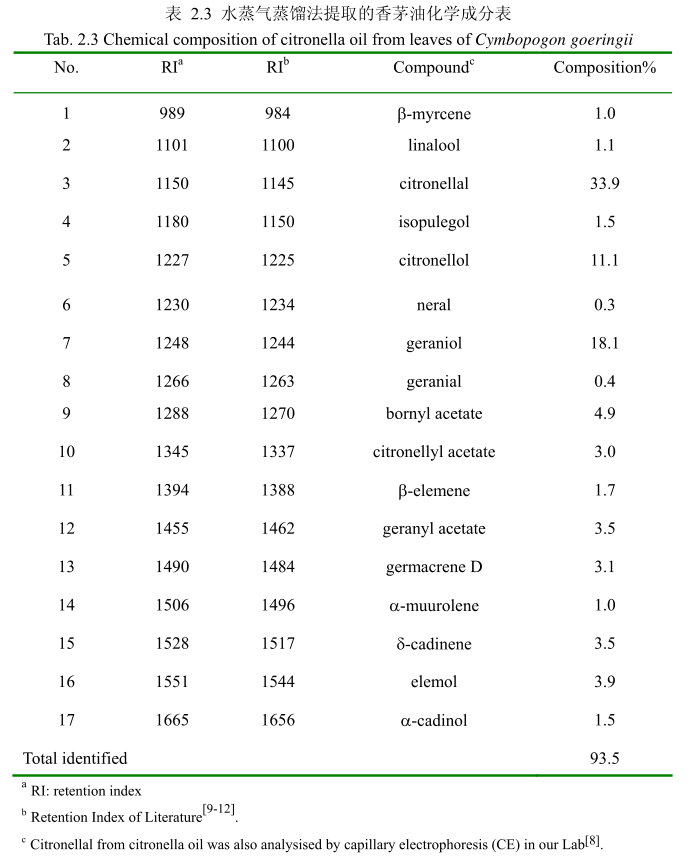
2.3.3.3.香茅油化学成分
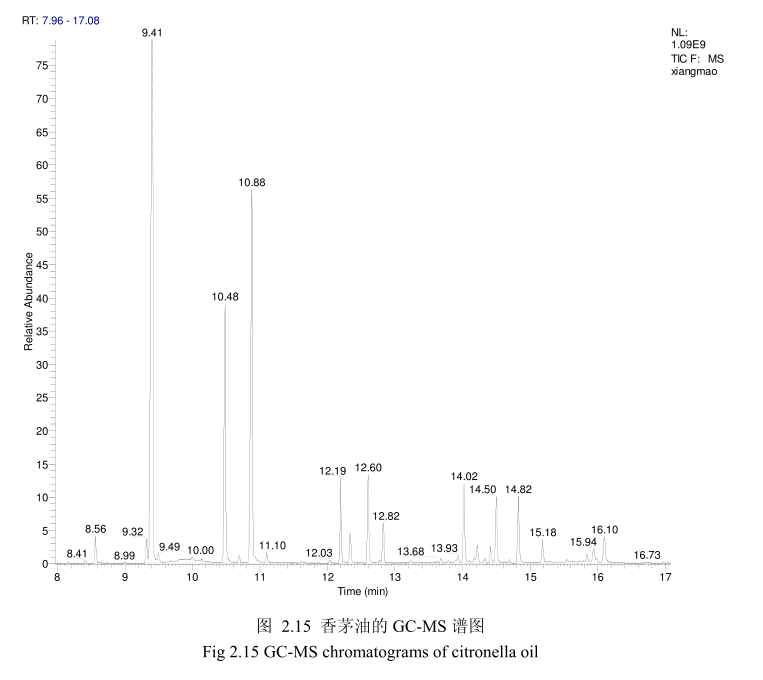
将最优提取条件下提取的香茅油通过GC-MS分析。图2.15是香茅油GC-MS图谱。从图2.15可以看出,香茅油的化学成分大多集中在保留时间6.0-17分钟之间。通过和标准品或质谱数据库数据比对,鉴定出17种化学成分,占总油含量的93.5%。表2.3为鉴定出的香茅油成分及含量。从表2.3可看出,其相对含量较高的成分为香茅醛(33.9%)、香叶醇(18.1%)和香茅醇(11.1%)等。
2.4 讨论
挥发油是一种成分非常复杂的天然混合物,通常含有20-60种成分,并且其含量差异很大。挥发油通常含有两种或三种主要特征成分,每种的含量可达20%-70%,而有些成分的含量非常少。通常情况下,挥发油的主要成分决定了挥发油的生物活性。挥发油的成分包含两类[13-16]。其中主要的一类由萜烯类和萜类化合物组成,另外一类由芳香族和脂肪族化合物组成,都属于小分子化合物。
萜类化合物是植物次生代谢产物,在自然界分布广泛,由异戊二烯单元组成的化合物及其衍生物,按照碳原子的数目可以分为单萜、倍半萜、二萜、三萜和多萜等。由于萜类化合物分子中具有不同的碳环数,因此又可分为链萜、单环萜、双环萜和三环萜等[17]。单萜由两个异戊二烯单元组成,是挥发油中最有代表性的分子,占挥发油组分的 90%,包含:桂叶烯、罗勒、萜品烯、水芹烯、香叶醇、芳樟醇香茅醇,、熏衣草醇、香橙醇、薄荷醇、松油醇、龙脑、小茴香醇、香叶醛、橙花醛香茅醛、薄荷酮、香芹酮、薄荷酮、胡椒酮、乙酸芳樟酯、乙酸异冰片酯、桉油素麝香草酚、香芹酚等。当分子具有旋光性,两种对映异构体常常存在于不同的植物中。
芳香化合物衍生于苯基丙烷,其种类少于萜类。萜类和芳香化合物在植物中合成路径是分开的,但在某些植物中是共用一条合成路径。芳香类化合物包括:桂皮醛、肉桂醇、丁香酚、茴香脑、肉豆蔻醚等
挥发油是一种混合物,其化学成分会因为植物物种、生长的地理环境、提取方法、采收时间等因素的差异而不同[18,19]。本章运用水蒸气蒸馏法提取的自然晾干的山苍子果实的挥发油呈淡黄色。GC-MS分析水蒸气蒸馏法提取的山苍子油鉴定出19种化学成分,占总油的92.4%。其主要成分柠檬醛的相对含量占57.4%,柠檬烯的含量占 8.1%,芳樟醇的含量为5.2%,B-月桂烯的相对含量为4.7%,a-松油醇的相对含量为3.9%,β-烯的相对含量为2.7%,α-蒎烯的相对含量为1.9%。本章运用 GC-MS 分析的山苍子油的主要化学成分与其它相关文献报道的山苍子油的主要化学成分基本一致[20,21],但是比例不同,从而证明天然挥发油含量的差异性[22]。例如,同一种类的山苍子油,其产地为美国,主要成分柠醛的相对含量为 85.7%,柠檬烯的相对含量为2.9%,芳樟醇的相对含量为1.7%。Wang等[23]研究的山苍子油中柠檬醛的含量为63.75%,柠檬烯的相对含量为7.38%,甲庚酮的相对含量为 3.54%,烯的相对含量为2.87%,4-伞花烃的含量为2.14%。
同样用GC-MS 分析产于中国广东的丁香花蕾油,鉴定出10种物质,占总油的 94.8%。主要成分丁香酚的相对含量为76.8%,乙酸丁香酯的相对含量为 9.5%,B-石竹烯的相对含量为6.0%。尽管其主要成分与其它文献报道的一致,但是其含量不一致。如产地为意大利的丁香花蕾油中丁香酚的相对含量为70.8%,B-石竹烯的相对含量为 24.9%,乙酸丁香酯的含量为4.2%。Moon 等[24]报道的丁香花蕾油中丁香酚的相对含量为76.8%,B-石竹烯的相对含量为17.4%,乙酸丁香酯的相对含量为 1.2%。
用GC-MS分析的香茅叶的挥发油鉴定出17种成分,占总油的93.5%。其中主要成分香茅醛的相对含量为33.9%、香叶醇的相对含量为18.1%、香茅醇的相对含量为11.1%。其主要成分与相关文献报道的香茅油的主要化学成分一致,但是其比例有一点差异。如中国云南产的香茅挥发油中主要成分香茅醛的含量为36.055%,香叶醇的含量为21.33%。
从本章分析的三种天然挥发油与其相关文献报道的结果相比较可以看出,尽管其主要化学成分一致,但是其含量有一定差异。而挥发油的化学成分及其含量决定了其生物活性,因此其挥发油的生物活性也可能存在较大差异。
2.5 本章小结
本章运用水蒸气蒸馏法萃取了产地中国广东的山苍子果实、丁香花蕾、香茅叶,获得挥发油的理化指标如下:
(1)山苍子油得率2.7%,澄清淡黄色液体;感官特征:具有清鲜、甜的果香,类似柠檬油的香气;相对密度0.889(20/20℃);折光指数1.482(20℃)。通过单因素实验,山苍子挥发油最佳提取工艺参数为山苍子粉碎至40目,浸泡时间8小时,料液比1:10,提取时间6小时。GC-MS分析山苍子油鉴定出19种化学成分,占总油的92.4%。其主要成分柠檬醛的相对含量占57.4%,柠烯的含量占8.1%,芳樟醇的含量为5.2%,B-月桂烯的相对含量为4.7%,a-松油醇的相对含量为3.9%,B-烯的相对含量为2.7%,a-的相对含量为1.9%。
(2)丁香花蕾油,得率3.5%,澄清淡黄色液体,辛香及丁香酚的特征香气;相对密度 1.045(20/20℃);折光指数15290(20℃)。通过单因素实验,丁香花蕾挥发油最佳提取工艺参数为丁香花蕾粉碎至40目,浸泡时间6小时,料液比1:12,提取时间6小时。用GC-MS 分析丁香花蕾油,鉴定出10种物质,占总油的94.8%。主要成分丁香酚的相对含量为76.8%,乙酸丁香酯的相对含量为9.5%,B-石竹烯的相对含量为6.0%。
(3)香茅油得率0.5%,澄清淡黄色液体;感官特征:具有轻快、新鲜的清甜果香,有成熟柠檬果皮的香气;相对密度0.889(20/20℃);折光指数 1.486(20℃)通过单因素实验,香茅叶挥发油最佳提取工艺参数为香茅粉碎至20目,浸泡时间4小时,料液比1:8,提取时间4小时。GC-MS 分析的香茅叶的挥发油鉴定出 17种成分,占总油的93.5%。其中主要成分香茅醛的相对含量为33.9%、香叶醇的相对含量为 18.1%、香茅醇的相对含量为11.1%。
2.6参考文献
[1] Masotti V, Juteau F, Bessie 're JM. Seasonal and phenological variations of the essential oil from the narrow endemic species Artemisia molinieri and its biological activities. Journal of Agricultural and Food Chemistry, 2003, 51:7115- -7121
[2] Jirovetz L, Buchbauer G, Stoilova I, et al. Chemical composition and antioxidant properties of clove leaf essential oil. Journal of Agricultural and Food Chemistry, 2006, 54(17):6303-6307
[3] Smith R, Cohen S, Doull J, et al. A procedure for the safety evaluation of natural flavor complexes used as ingredients in food: essential oils. Food and Chemical Toxicology, 2005, 43(3):345-363
[4]Huang B, Ban X, He J, et al. Comparative analysis of essential oil components and antioxidant activity of extracts of Nelumbo nucifera from various areas of China. Journal of Agricultural and Food Chemistry, 2009, 58(1):441-448
[5]Wang Q, Yang Y, Zhao X, et al. Chemical variation in the essential oil of Ephedra sinica from Northeastern China. Food Chemistry, 2006, 98( 1):52-58
[6] Wang HW, Liu YQ. Chemical composition and antibacterial activity of essential oils from different parts of Litsea cubeba. Chemistry & Biodiversity, 2010,7:229-235.
[7]Mockute D, Bernotiene G. The main citral-geraniol and carvacrol chemotypes of the essential oil of Thymus Pulegioides L. growing wild in Vilnius District (Lithuania). Journal of Agricultural and Food Chemistry,1999, 47: 3787-3790
[8]Huang XW, Yi L,Gao ZH, et al. Determination of seven active ingredients in three plant essential oils by using micellar electrokinetic chromatography. Analytical Letters, 2012,45, 2014-2015
[9]Jirovetz L, Buchbauer G, Stoilova I, et al. Chemical composition and antioxidant properties of clove leaf essential oil. Journal of Agricultural and Food Chemistry, 2006, 54, 6303-6307
[10] Kiran SR, Reddy AS, Devi PS,et al. Reddy, Insecticidal, antifeedant and oviposition deterrent effects of the essential oil and individual compounds from leaves of Chloroxylon swietenia DC. Pest Management Science, 2006, 62:1116-1121
[11] Lota ML, Serra DD, Tomi F,et al. Volatile components of peel and leaf oils of Lemon and Lime Species. Journal of Agricultural and Food Chemistry, 2002, 50:796-805
[12] Rajeswara R, Bhattacharya AK, Mallavarapu GR,et al. Yellowing and crinkling disease and its impact on the yield and composition of the essential oil of citronella (Cymbopogon winterianus Jowitt). Flavour Fragrance Jourmal, 2004, 19:344-350
[13]Da Silva EB, Matsuo AL, Figueiredo CR, et al. Chemical constituents and cytotoxic evaluation of essential oils from leaves of Porcelia macrocarpa(Annonaceae). Natural Product Communications, 2013, 8(2):277-279
[14]Betts TJ. Chemical characterisation of the different types of volatile oil constituents by various solute retention ratios with the use of conventional and novel commercial gas chromatographic stationary phases. Journal ofChromatography A, 2001, 936(1):33-46
[15]Setzer WN. Essential oils and anxiolytic aromatherapy. Natural Product Communications,2009,4(9): 1305-1316.
[16] Pichersky E, Noel JP, Dudareva N. Biosynthesis of plant voltiles: nature's diversity and ingenuity. Science Signaling, 2006, 311(5762):808-811
[17] 张长波,孙红霞,巩中军.植物萜类化合物的天然合成途径及其相关合酶.植物生理学通讯,2007,43(4) :779-785
[18] Lota ML, De Rocca Serra D, Tomi F, et al. Volatile components of peel and leaf oils of lemon and lime species. Journal of Agricultural and Food Chemitry, 2002, 50(4):796-805
[19] Lalko J, Api A. Investigation of the dermal sensitization potential of various essential oils in the local lymph node assay. Food and Chemical Toxicology, 2006, 44(5):739-746
[20] Gao Z, Xu H, Chen X, et al. Antioxidant status and mineral contents in tissues of rutin and baicalin fed rats. Life Sciences, 2003, 73(12):1599-1607
[21] Ho CL, Jie-Pinge O, Liu YC, et al. Compositions and in vitro anticancer activities of the leaf and fruit oils of Litsea cubeba from Taiwan. Natural Product
Communications, 2010, 5(4):617-620
[22] Liu ZL, Liu QR, Chu SS, et al. Insecticidal activity and chemical composition of the essential oils of Artemisia lavandulae folia and Artemisia sieversiana from China. Chemistry & Biodiversity, 2010, 7(8):2040-2045
[23] Wang H, Liu Y. Chemical composition and antibacterial activity of essential oils from different parts of Litsea cubeba. Chemistry & Biodiversity, 2010,
7(1):229-235
[24] Moon SE, Kim HY, Cha JD. Synergistic effect between clove oil and its major compounds and antibiotics against oral bacteria. Archives of Oral Biology, 2011 ,56(9):907-916
未完待续……
摘 要
随着大气层中臭氧的逐渐减少,地球表面的紫外线强度不断增加。高强度的紫外照射会引起皮肤色素的过度沉着和皮肤功能的紊乱。因此研发有效的祛斑、美白护肤品一直是化妆品领域的热门之一。近年来,出于安全性方面的考虑,人们更愿意接受源自天然中草药的美白护肤产品。中国是一个植物物种多样的国家,有许多药食同源的植物挥发油在民间被广泛地应用于护肤品,但其是否具有祛斑、美白的功能,却鲜有研究。本论文选取了三种各具独特功能的本土植物挥发油(丁香花蕾油、香茅油和山苍子油),通过系列实验,为判断其是否具有成为美白护肤品成分的潜能提供科学依据。
主要内容包含以下几个方面:
1、优化了提取山苍子油、丁香花蕾油和香茅油的提取工艺,并用气质联用技术鉴定了三种挥发油的主要成分。鉴定出山苍子油中的19种化学成分,占整个挥发油组分的 92.4%。主要成分有柠檬醛(57.4%),柠檬烯(8.1%),芳樟醇(5.2%),B-月桂烯(4.7%),α-松油醇(3.9%),β-蒎烯(2.7%)和a-蒎烯(1.9%);鉴定出丁香花蕾油中的 10种成分,占整个挥发油的94.8%。主要成分有丁香酚(76.8%),乙酸丁香酯(9.5%)和B-石竹烯(6.0%);鉴定出香茅油中的 17 种成分,占整个挥发油的 93.5%。主要成分有香茅醛(33.9%),香叶醇(18.1%),和香茅醇(11.1%)。
2、通过酪氨酸酶抑制试验来评价三种挥发油及其主要成分对酪氨酸酶的抑制效果。结果表明山苍子油和其主要成分柠檬醛对酪氨酸酶单酚酶和二酚酶活性有抑制能力,对酪氨酸酶二酚酶的抑制能力强于酪氨酸酶单酚酶,且动力学结果表明柠檬醛对酪氨酸酶是非竞争性抑制作用。丁香花蕾油和丁香酚对酪氨酸酶单酚酶具有一定的抑制作用,但是对酪氨酸酶二酚酶没有表现出任何抑制作用。而香茅油及其主要成分对酪氨酸酶没有抑制作用。
3、分别研究了山苍子油、香茅油、丁香花蕾油和其主要成分的抗氧化活性。
结果表明丁香花蕾油具有最强的自由基清除能力(超阴离子的ICo值为58g/mL;ABTS的ICso值为8.5μg/mL)和强的抑制脂质过氧化的能力;香茅油的自由基清除能力和抑制脂质过氧化的能力低于丁香花蕾油(超氧阴离子的ICso值为150 μg/mL;ABTS*的ICso值为 500μg/mL)。山苍子油的自由基清除能力最低(超氧阴离子的ICso值为10.2mg/mL;ABTS 的ICso值为 17.75 mg/mL)。
4、为了探讨山苍子油、丁香花蕾油和香茅油分别对 UV-TiO2-NO,体系导致的蛋白质氧化和酪氨酸硝化损伤的抑制作用,将牛血清蛋白作为模型蛋白进行体外试验,采用免疫印迹法测定牛血清白蛋白的氧化和酪氨酸硝化。结果表明丁香花蕾油、香茅油和山苍子油对UV-TiO,-NO,-催化的蛋白质氧化和酪氨酸硝化都有较强的抑制效果且呈剂量依耐性。
关键词:挥发油,酪氨酸酶,抗氧化,纳米二氧化钛,蛋白质氧化,蛋白质酪氨酸硝化
Abstract
Ultraviolet (UV) radiation on the surface of the earth is certainly increasing as a result of the depletion of stratospheric ozone. High-intensity or accumulated UV radiation provokes many hyperpigmentation disorders, which are of particular concern to women as well as men, In recent years, the study of traditional herbal medicines for skin-protecting and skin-lighting has become a hot spot in the cosmetics field, due to various safety concerns and interesting largely unexplored sources. In this dissertation. the main objective was to investigate the potential usage of clove oil , citronella oil and Litsea cubeba oil as natural sources of tyrosinase inhibitor, antioxidant, and protectionprotein against UVTiO,-NO2- induced protein oxidation and tyrosine nitration in jury The main results are as follows:
1. The clove oil, citronella oil and Litsea cubeba oil extracted by hydrodistillation, showed light yellow in color. The total of 10 compounds was identified by GC-MS in the clove oil, accounting for 94.8% of the total oil. The main components were eugenol(76.8%), followed by eugenyl acetate (9.5%) and β-caryophyllene (6.0%). Essential oil from Cymbopogon goeringii leaves was also analyzed by GC-MS, resulted in the identification of 17 compounds representing 93.5% of the oil. Citronellal (33.9%),geraniol (18.1%), and citronellol (11.1%) were the main compounds. GC-MS analyses of Litsea cubeba oil led to identification of 19 different compounds, accounting for 92.4%of the total oil. The main components were citral representing 57.4% of the total amount of oil extracted, follow by limonene(8.1%),linalool (5.2%),β-myrcene (4.7%),a-terpineol (3.9%),β-pinene(2.7%) and a-pinene(1.9%%).
2. Tyrosinase inhibitory assay was used to evaluate whether the three essential oilsand their main components possessed potential inhibitory effect on tyrosinase. The results show that Litsea cubeba oil and citral exhibited inhibitory effect on monophenolase and diphenolase activity of tyrosinase. The inhibitory effect of Litseacubeba oil and citral on diphenolase activity was stronger than monophenolase activity oftyrosinase. The inhibition pattern of mushroom tyrosinase by citral was determined byLineweaver-Burk polt analysis. The results demonstrated that citral was anoncompetition tyrosinase inhibitor, Clove oil and eugenol exhibited some inhibitoryeffect on monophenolase activity of` tyrosinase, but not show any inhibitory effect on diphenolase activity of tyrisinase. Citronella oil and their main components didn'texhibited any inhibitory effect.
3. In order to determine whether the three essential oils and their main components had antioxidant activity, ABTS cation radical scavenging assay, superoxide anion radicalscavenging assay and lipid peroxidation assay were carried out. Clove oil exhibited prominent radical scavenging activities (ICso: 58 μg/ mL for O2 and 8.5 ug/ mL for ABTS*) and strong inhibitory effect on lipid peroxidation, whilst citronella oil showedweaker antioxidative activities (ICso: 150 μg/ mL for O, and 500 μg/ mL for ABTS )and lipid peroxidation inhibitory efficiency. Litsea cubeba oil exhibited the lowest anti-oxidative activities (ICso: 17.75 mg/mL for ABTS , 10.2 mg/mL, for O, ).
4. In order to investigate inhibitory effects of essential oils to UV-TiO,-NOzinduced protein oxidation and protein tyrosine nitration injury, bovine serum albumin(BSA) were used as model protein, inhibition were followed by $DS-PAGE and Western blotting methods. The results showed that clove oil, citronella oil and Litsea cubeba oil exhibited satisfactory protective effects and revealed dose-dependence in protecting BSAagainst UV - TiO, - NO,` induced protein oxidation and tyrosine nitration injury.
Keywords: essential oil, tyrosinase, antioxidant, nano TiO2, protein oxidation, tyrosine nitration



