文献精选




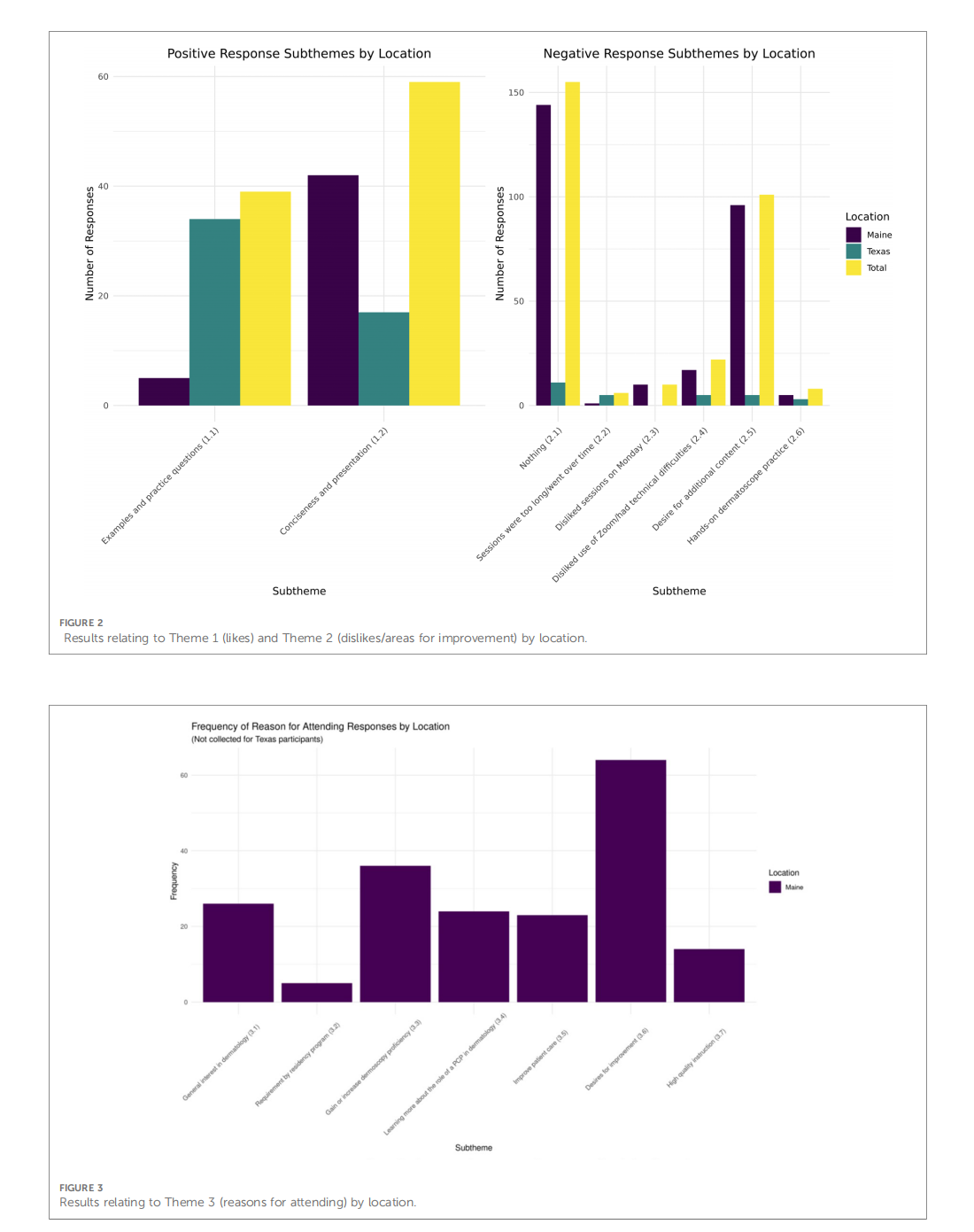


This article is excerpted from the 《Frontiers in Digital Health》 by Wound World






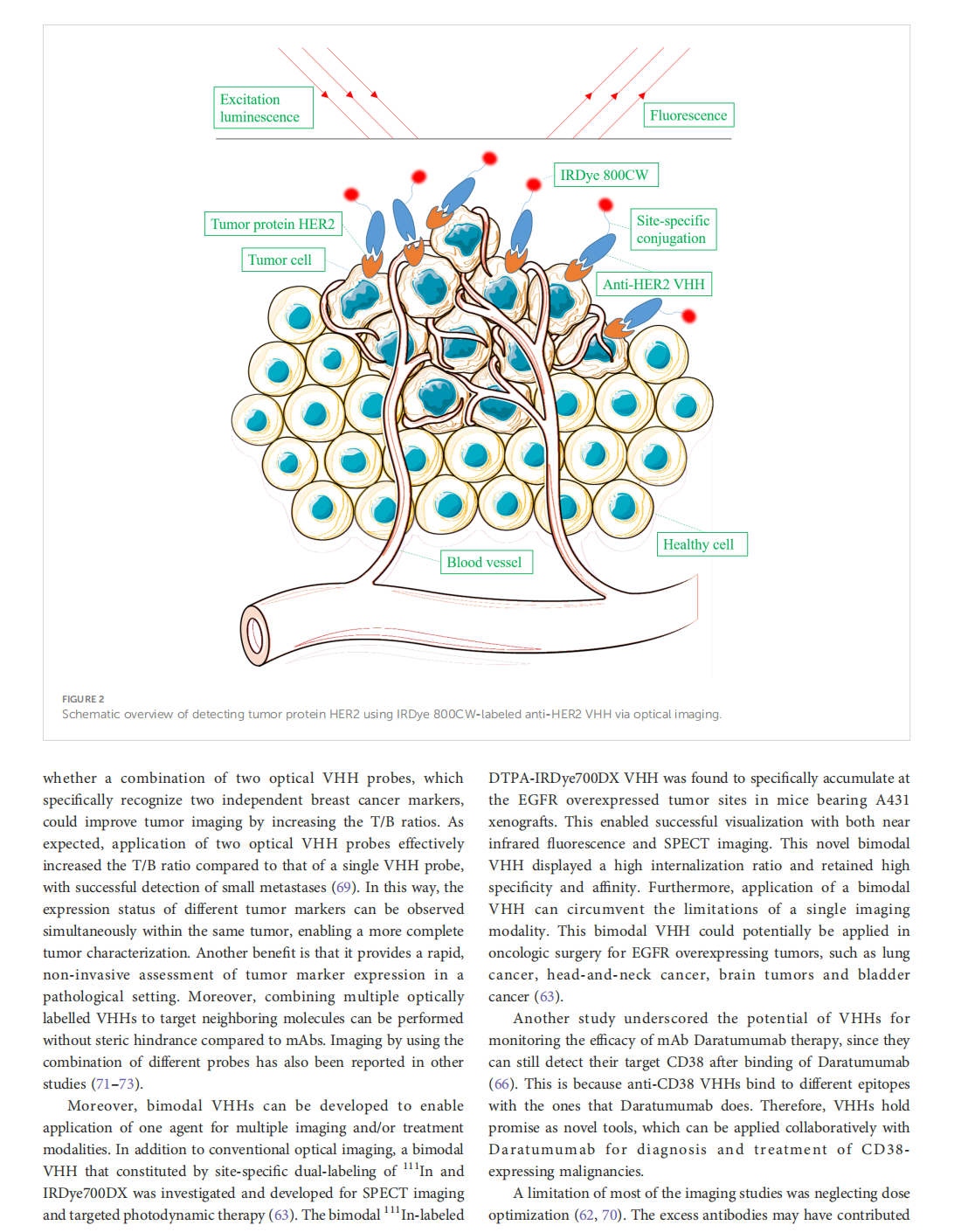











This article is excerpted from the 《Frontiers in Oncology》 by Wound World








This article is excerpted from the《Frontiers in Medicine》 by Wound World






This article is excerpted from the 《Frontiers in Aging》 by Wound World




